Incidence and predictors of all-cause mortality within one year after adult spinal deformity surgery
Introduction
In adults with degenerative scoliosis and back pain, adult spinal deformity (ASD) surgery offers significant reduction in pain and improved quality of life relative to medical intervention (1-5). Various surgical options exist to optimize functional outcomes while limiting morbidity. However, the risk of perioperative complications remains high (6). Although the incidence of adverse events varies based on outcomes criteria, prior studies have endorsed complication rates as high as 70% (2,7). Approximately 15–20% of ASD patients may also require reoperation (8). ASD surgery has also been associated with mortality in the postoperative period, secondary to complications such as sepsis or pulmonary embolism (9). Mortality rates are reported to range from 0.03% to 3.52% for this surgical population (9) (10,11).
Due to the established morbidity following ASD surgery, risk stratification is paramount for optimal patient selection. Several prior analyses have identified risk factors for postoperative complications following ASD surgery (6,12); however, predictive models of all-cause mortality have not been adequately explored, likely due to the rarity of this event. Though anesthetic risk score has been previously linked to mortality (9), specific demographic and perioperative factors that independently contribute to the risk of death have not been identified. Moreover, prior studies have assessed death at 30 days, rather than longer postoperative periods. Knowing which risk factors are associated with all-cause mortality could improve patient selection, optimize perioperative resource allocation, and mitigate poor outcomes.
We sought to utilize a large, prospective database to evaluate all-cause mortality within 1 year of ASD surgery. The objective of the current study was to report the incidence of all-cause mortality within 1 year following ASD surgery along with preoperative factors and postoperative complications predictive of this outcome.
Methods
Patient identification
The 2008–2014 American College of Surgeons National Surgical Quality Improvement Program (ACS-NSQIP) database was used to collect data for patients who underwent ASD surgery. Over 600 hospitals participate in the NSQIP registry, and trained, audited data collection specialists prospectively record information in accordance with rigorous definitions (13). NSQIP collects roughly 135 clinical variables on patients undergoing major surgeries from the preoperative, operative, and postoperative time periods within 30 days of the index operation, in addition to certain longer term outcomes, including death (13). The case series for the present study was generated by employing the following Current Procedural Terminology (CPT) codes:
- 22800 (posterior arthrodesis for spinal deformity up to 6 segments);
- 22802 (posterior arthrodesis for spinal deformity 7 to 12 segments);
- 22804 (posterior arthrodesis for spinal deformity 13 or more segments);
- 22808 (anterior arthrodesis for spinal deformity 2 to 3 segments);
- 22810 (anterior arthrodesis for spinal deformity 4 to 7 segments);
- 22812 (anterior arthrodesis for spinal deformity 8 or more segments);
- 22818 (kyphectomy, resection of vertebral segment, single or 2 segments);
- 22819 (kyphectomy, resection of vertebral segment, 3 or more segments);
- 22843 (posterior segmental instrumentation, 7 to 12 segments);
- 22844 (posterior segmental instrumentation, 13 or more segments);
- 22846 (anterior instrumentation, 4 to 7 segments);
- 22847 (anterior instrumentation, 8 or more segments).
Arthrodesis codes such as 22610 (posterior arthrodesis, single level thoracic), 22612 (posterior arthrodesis, single level lumbar), 22830 (exploration of spinal fusion), and 22633 (posterior arthrodesis, interbody technique, single lumbar interspace) were considered inherent within the use of the instrumentation codes in ASD. Patients with CPT codes 22842 (posterior segmental instrumentation, 3 to 6 segments) or 22845 (anterior instrumentation, 2 to 3 segments) were also included if they were diagnosed with spinal deformity according to an International Classifications of Diseases (ICD-9) code of 737.1, 737.2, 737.3, 737.4, 737.8, or 737.9. These coding parameters were based on previous NSQIP studies of this surgical population (14-16).
Data collection
Outcome
The primary outcome of interest was death within 1 year following ASD surgery. Since the direct cause of death could not be obtained, the principle outcome represented the event of all-cause mortality. This outcome was created by analyzing the variables “year of surgery” and “year of death”—patients who had the same year for both variables were classified as death within 1 year following ASD surgery.
Preoperative
Selected preoperative demographic and past medical history variables relevant to the outcome of death included: sex, age, body mass index (BMI), American Society of Anesthesiologists (ASA) score, hypertension (requiring medication), diabetes (requiring an oral-agent or insulin for glucose control), cardiac history (previous stent, cardiac surgery, or angina), dialysis due to chronic kidney disease, prior cerebrovascular accident or transient ischemic attack (CVA/TIA), history of cancer, open wound, steroid use, bleeding disorder (chronic condition that places patient at risk for excessive bleeding such as vitamin K deficiency, hemophilia, or continued anticoagulation therapy), ventilator use (ventilator-assisted respiration in 48 h before surgery), and rapid weight loss (unintentional >10% weight loss in last 6 months), Additional social history factors included: cigarette use (in last 1 year) and functional dependency (independent versus partially or totally dependent).
Postoperative
Selected postoperative complications relevant to the outcome of death were classified as major or minor in accordance with prior neurosurgical NSQIP studies (15-17). Minor complications included superficial or deep surgical site infection (SSI), pneumonia, urinary tract infection (UTI), and pneumonia. Major complications included deep venous thrombosis (DVT), acute kidney injury (AKI), ventilator for >48 h, unplanned intubation, sepsis/septic shock, pulmonary embolism (PE), acute CVA, or myocardial infarction (MI).
Statistical analysis
Descriptive statistics were compiled for all demographics, preoperative, and postoperative characteristics. Cases with missing data for any of the variables were excluded via a list-wise deletion method. For the preoperative factor section, univariate logistic regression analyses were performed to test the association between each variable and the primary outcome of death within 1 year. Variables that resulted in an association of P<0.10 were then incorporated into a multivariable logistic regression model to identify the independent predictors of death. For the postoperative complication section, due to the number of preoperative covariates associated with the outcome of death, a propensity score analysis was used to avoid an unstable model (18). The propensity score was defined as the probability that an individual would have a complication based on the aforementioned preoperative factors: sex, age, BMI, PMH, and SH. Using each propensity score, a similar process of univariate logistic regression analyses followed by multivariable logistic regression was completed to identify the independent predictors of the primary outcome of death within 1 year. Regardless of univariate P value, all major complications were included in the final multivariable models due to prior research demonstrating the importance of each major complication. All statistical analyses were performed in STATA version 14 (StataCorp LP, College Station, Texas).
Results
Preoperative and postoperative variables
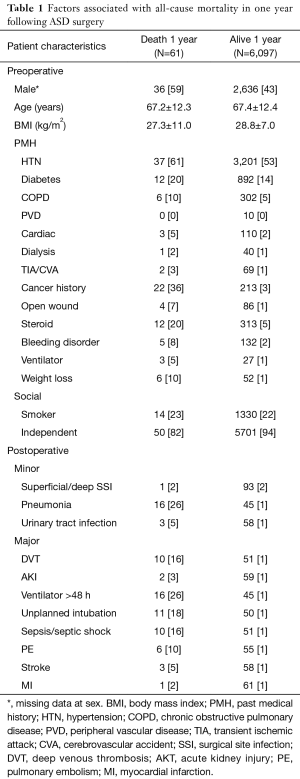
A total of 6,158 patients surgically treated for ASD from the NSQIP database met inclusion criteria. Of these, 61 (0.99%) died within one year of the index ASD surgery. Preoperative variables and postoperative complications are summarized in Table 1.
Full table
Preoperative predictors of death
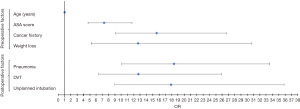
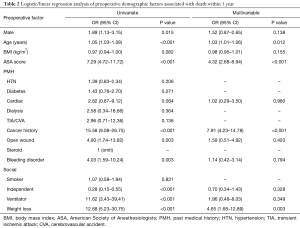
Results depicting the preoperative predictors via univariate and multivariable logistic regression modeling are presented (Table 2, Figure 1). Preoperative variables that resulted in P<0.10 in association with all-cause mortality included: male sex, increasing age, BMI, ASA score, cardiac history, cancer history, open wound, bleeding disorder, functional living status, ventilator dependency, and unexpected weight loss. In the multivariable model controlling for age, gender, ASA score and postoperative complications, four independent risk factors emerged: increasing age (OR =1.03; 95% CI, 1.01–1.06, P=0.012), ASA score (OR =4.32; 95% CI, 2.68–6.94; P<0.001), cancer history (OR =7.91; 95% CI, 4.23–14.78; P<0.001) and unintentional weight loss (OR =4.65; 95% CI, 1.68–12.89; P=0.003).
Full table
Postoperative predictors of death
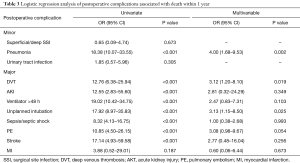
Results of the postoperative complications univariate and multivariable logistic regression models are seen (Table 3, Figure 1). Postoperative complications that resulted in P<0.10 in association with all-cause mortality included: pneumonia, DVT, AKI, ventilator for greater than 48 hours, unplanned intubation, sepsis/septic shock, PE, stroke, and MI. After propensity score matching and multivariable logistic regression, controlling for all major postoperative complications, three independent risk factors emerged: pneumonia (OR =4.00; 95% CI, 1.68–9.53), DVT (OR =12.76; 95% CI, 1.20–8.10) and unplanned intubation (OR =3.13; 95% CI, 1.15–8.50).
Full table
Discussion
While predictors of morbidity after ASD surgery have been well studied, less is known about the independent predictors of mortality. We thus sought to quantify the incidence and factors associated with all-cause mortality within 1 year following ASD surgery. The following preoperative factors conferred an independent risk of the death within 1 year, including increasing age, increasing ASA score, cancer history, and unintentional weight loss, along with the postoperative complications of pneumonia, DVT, and unplanned intubation. Though the ultimate cause of death remains unknown, the current predictive analysis of all-cause mortality within a 1-year following ASD surgery may have additional implications for the preoperative, intraoperative, and postoperative management of ASD patients.
The incidence of mortality following ASD surgery has been reported in several studies across varying follow-up periods. Several authors have reported that death occurs in less than 1% of those undergoing ASD surgery, including mortality rates of 0.28% and 0.4% (10,11,19,20). A multi-institution, prospective registry endorsed a 1.2% mortality rate at 2 years (2). Additionally, a single institution study of 361 patients published a 2.4% mortality risk within the 30-day postoperative period (9). Causes of mortality included pulmonary embolism, sepsis, shock, and MI. Though cause of death could not be determined, our study demonstrated a 1.0% all-cause mortality rate within a 1-year period, which appears to align with the overall incidence in prior reports.
Prior to a discussion of risk factors, it must be emphasized that specific cause of death could not be determined from the current data. All-cause mortality within 1 year was the primary outcome of interest. It is possible that the cause of death may not be directly related to the prior ASD surgery. In these cases, it can be deduced that mortality was reflective of the decreased health status and comorbidities of the ASD population.
Preoperative risk stratification
The current study revealed four significant preoperative variables associated with all-cause mortality, which may have a role in surgical risk stratification. The association between ASA score and 30-day mortality has been identified in prior studies of ASD surgery, in which ASA score was significantly different between groups that did or did not experience postoperative death (9). This observation was reaffirmed in our results when the follow-up period was extended to 1 year. Attention should thus be paid to the level of anesthetic risk of each patient, as ASA class 3 and 4 patients are at inherently greater risk. However, it must be noted that reliability of ASA score varies between anesthesiologist, creating a need for more objective, quantitative methods for preoperative risk assessment. Another alternate risk score is the modified frailty index (mFI), a composite scoring system of 11 variables, including diabetes, functional status, pneumonia, among others (21). Though not studied in our analysis, Leven and co-authors (10) evaluated 1,001 patients undergoing ASD surgery and found that increasing mFI score was associated with increased risk of mortality. For increasing levels of mFI, mortality risk was 0.3%, 1.6%, 0.8%, 10.0%, and 0.0%, which demonstrates the non-linear trend and need for further scrutiny. Additional comorbidity systems include the Charlson Comorbidity Index (22), a composite score based on 17 weighted comorbidities, and the Elixhauser comorbidity system (23), a set of 30 indicators without a composite measure. These preoperative scoring systems may help quantify risk based on objective comorbidity profiles and improve upon the variable ASA score.
Age was also a significant factor in our study, which aligns with prior findings (11). However, a healthy 70-year-old patient may be a better ASD surgery candidate than an unhealthy 50-year-old patient, and the current authors advocate for age to be taken in context of overall health status. Cancer history and unintended weight loss were also found to be predictive factors of death within one year. Due to data constraints, we were unable to distinguish between active cancer versus remission, nor whether patients were undergoing cancer treatment at the time of surgery. Thus, any history of cancer should be evaluated immediately prior to ASD surgery with an independent oncologist, and the use of BMP in these patients should be avoided (24). Since these factors are known prior to surgery, these preoperative factors should help determine the operative plan and appropriate level of surgical invasiveness, ranging from decompression alone, to combined anterior and posterior approaches and major osteotomies (25).
Postoperative management
The three independent predictors of death in the postoperative period were pneumonia, DVT, and unplanned intubation. Pneumonia is a common inpatient complication, and a number of preventative measures have been evaluated for respiratory complications (26,27). Wren et al. (27) observed a significant decrease in rates of pneumonia in a cohort of postoperative non-cardiac, surgical patients through the implementation of a pneumonia-prevention program, which included the use of incentive spirometry (25). Though PNA itself is not often fatal, a possible explanation for this association is that PNA may lead to downstream effects such as decreased mobility, cardiovascular strain, and lower functional status, which in turn increase the likelihood of death. Due to the grave implications of pneumonia in postsurgical ASD patients, techniques for preventing respiratory infection should be considered.
The risks and prevention of DVT have also been studied extensively, particularly in spine surgery (28). Patients with a known coagulopathy should have more aggressive anticoagulation approaches taken, along with those undergoing anterior approaches and retraction of iliac vessels. Chemical prophylaxis may not be necessary with a posterior-only approach and early mobilization; however, in less mobile patients, chemical prophylaxis can be started on postoperative day one. The association between mortality and unplanned intubation has been previously investigated in an ASD population. De la Garza et al. (19) found that the mortality rate for patients who underwent unplanned intubation was 7.3%, significantly higher than controls. This may be partially explained because the conditions that require unplanned intubation, such as respiratory failure or sepsis, may also increase mortality risk. However, these authors found that unplanned intubation was independently predictive of mortality while controlling for various perioperative factors. This corroborates our findings, and demonstrates that it is particularly important to manage conditions that lead to intubation and maintain close communication with intensivists at all times.
It should also be noted that the more severe complications of sepsis, stroke, or MI were not associated with death. Though logic would suggest that these complications could increase the chance of death, these findings may be explained by the low numbers in both cohorts. When assessing correlation between two exceedingly rare events, it is possible that accurate correlations are lost. Thus, we must underscore the importance of these additional complications despite a lack of statistical significance. Regardless, the complications identified in this study seem to particularly augment the risk of mortality for this surgical population. Close surveillance and prophylactic measures should be considered for these patients.
Limitations
This study is limited in several capacities. The analysis of this prospectively collected dataset was performed retrospectively, which presents an intrinsic risk of selection bias. Additionally, the methodology used to extract 1-year mortality data underestimated the number of deaths due to data collection artifacts. For instance, a surgery occurring in the latter half of 1 year that resulted in death the following calendar year would be excluded from analysis. Furthermore, exact cause of death could not be obtained from the current database, and in place, only all-cause mortality was studied. Thus, the exact relation of death to ASD surgery could not be determined. There were also pertinent details regarding specific comorbidities, such as cancer status and treatment course, that were not available in the NSQIP database. Lastly, given the rarity of some postoperative complications, it is possible an unstable logistic regression model was created due to the number of covariates included.
Conclusions
Perioperative death attributed to surgical invention for ASD is a rare complication, for which risk factors have not been adequately explored. There appear to be specific comorbidities, namely age, ASA score, cancer history, and unintentional weight loss that predispose patients to postoperative mortality. Additionally, complications such as pneumonia, DVT, and unplanned intubation, seem more likely to fatally progress for these patients. These factors associated with all-cause mortality may have implications for planning the appropriate treatment course and operative intervention for this patient population, as well as for prophylactic management of postoperative complications.
Acknowledgements
The authors would like to acknowledge the NSQIP database personnel for their rigorous data collection methods and public use of their database.
Footnote
Conflicts of Interest: previous presentations—this abstract was presented at the CNS/AANS Spine Section 2018 in Orlando, FL.
Ethical Statement: Vanderbilt IRB was exempted for the study.
References
- Scheer JK, Smith JS, Clark AJ, et al. Comprehensive study of back and leg pain improvements after adult spinal deformity surgery: analysis of 421 patients with 2-year follow-up and of the impact of the surgery on treatment satisfaction. J Neurosurg Spine 2015;22:540-53. [Crossref] [PubMed]
- Ames CP, Scheer JK, Lafage V, et al. Adult Spinal Deformity: Epidemiology, Health Impact, Evaluation, and Management. Spine Deform 2016;4:310-22. [Crossref] [PubMed]
- Smith JS, Klineberg E, Lafage V, et al. Prospective multicenter assessment of perioperative and minimum 2-year postoperative complication rates associated with adult spinal deformity surgery. J Neurosurg Spine 2016;25:1-14. [Crossref] [PubMed]
- Smith JS, Shaffrey CI, Berven S, et al. Improvement of back pain with operative and nonoperative treatment in adults with scoliosis. Neurosurgery 2009;65:86-93; discussion 93-4. [Crossref] [PubMed]
- Yoshida G, Boissiere L, Larrieu D, et al. Advantages and Disadvantages of Adult Spinal Deformity Surgery and Its Impact on Health-Related Quality of Life. Spine (Phila Pa 1976) 2017;42:411-9. [Crossref] [PubMed]
- Schwab FJ, Hawkinson N, Lafage V, et al. Risk factors for major peri-operative complications in adult spinal deformity surgery: a multi-center review of 953 consecutive patients. Eur Spine J 2012;21:2603-10. [Crossref] [PubMed]
- Yadla S, Maltenfort MG, Ratliff JK, et al. Adult scoliosis surgery outcomes: a systematic review. Neurosurg Focus 2010;28. [Crossref] [PubMed]
- Scheer JK, Tang JA, Smith JS, et al. Reoperation rates and impact on outcome in a large, prospective, multicenter, adult spinal deformity database: clinical article. J Neurosurg Spine 2013;19:464-70. [Crossref] [PubMed]
- Pateder DB, Gonzales RA, Kebaish KM, et al. Short-term mortality and its association with independent risk factors in adult spinal deformity surgery. Spine (Phila Pa 1976) 2008;33:1224-8. [Crossref] [PubMed]
- Leven DM, Lee NJ, Kothari P, et al. Frailty Index Is a Significant Predictor of Complications and Mortality After Surgery for Adult Spinal Deformity. Spine (Phila Pa 1976) 2016;41:E1394-401. [Crossref] [PubMed]
- Worley N, Marascalchi B, Jalai CM, et al. Predictors of inpatient morbidity and mortality in adult spinal deformity surgery. Eur Spine J 2016;25:819-27. [Crossref] [PubMed]
- Pull ter Gunne AF, Cohen DB. Incidence, prevalence, and analysis of risk factors for surgical site infection following adult spinal surgery. Spine (Phila Pa 1976) 2009;34:1422-8. [Crossref] [PubMed]
- Shiloach M, Frencher SK Jr, Steeger JE, et al. Toward robust information: data quality and inter-rater reliability in the American College of Surgeons National Surgical Quality Improvement Program. J Am Coll Surg 2010;210:6-16. [Crossref] [PubMed]
- Di Capua J, Somani S, Kim JS, et al. Hospital-Acquired Conditions in Adult Spinal Deformity Surgery: Predictors for Hospital-Acquired Conditions and Other 30-Day Postoperative Outcomes. Spine (Phila Pa 1976) 2017;42:595-602. [Crossref] [PubMed]
- Kothari P, Lee NJ, Lakomkin N, et al. Impact of Resident Involvement on Morbidity in Adult Patients Undergoing Fusion for Spinal Deformity. Spine (Phila Pa 1976) 2016;41:1296-302. [Crossref] [PubMed]
- Kothari P, Lee NJ, Leven DM, et al. Impact of Gender on 30-Day Complications After Adult Spinal Deformity Surgery. Spine (Phila Pa 1976) 2016;41:1133-8. [Crossref] [PubMed]
- Kothari P, Somani S, Lee NJ, et al. Thirty-Day Morbidity Associated with Pelvic Fixation in Adult Patients Undergoing Fusion for Spinal Deformity: A Propensity-Matched Analysis. Global Spine J 2017;7:39-46. [Crossref] [PubMed]
- Seeger JD, Kurth T, Walker AM. Use of propensity score technique to account for exposure-related covariates: an example and lesson. Med Care 2007;45:S143-8. [Crossref] [PubMed]
- De la Garza Ramos R, Passias PG, Schwab F, et al. Incidence, Risk Factors, and Mortality of Reintubation in Adult Spinal Deformity Surgery. Clin Spine Surg 2017;30:E896-900. [Crossref] [PubMed]
- De la Garza-Ramos R, Jain A, Kebaish KM, et al. Inpatient morbidity and mortality after adult spinal deformity surgery in teaching versus nonteaching hospitals. J Neurosurg Spine 2016;25:15-20. [Crossref] [PubMed]
- Saxton A, Velanovich V. Preoperative frailty and quality of life as predictors of postoperative complications. Ann Surg 2011;253:1223-9. [Crossref] [PubMed]
- Charlson ME, Pompei P, Ales KL, et al. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis 1987;40:373-83. [Crossref] [PubMed]
- Elixhauser A, Steiner C, Harris DR, et al. Comorbidity measures for use with administrative data. Med Care 1998;36:8-27. [Crossref] [PubMed]
- Carragee EJ, Chu G, Rohatgi R, et al. Cancer risk after use of recombinant bone morphogenetic protein-2 for spinal arthrodesis. J Bone Joint Surg Am 2013;95:1537-45. [Crossref] [PubMed]
- Silva FE, Lenke LG. Adult degenerative scoliosis: evaluation and management. Neurosurg Focus 2010;28. [Crossref] [PubMed]
- Westwood K, Griffin M, Roberts K, et al. Incentive spirometry decreases respiratory complications following major abdominal surgery. Surgeon 2007;5:339-42. [Crossref] [PubMed]
- Wren SM, Martin M, Yoon JK, et al. Postoperative pneumonia-prevention program for the inpatient surgical ward. J Am Coll Surg 2010;210:491-5. [Crossref] [PubMed]
- Edwards CC 2nd, Lessing NL, Ford L, et al. Deep Vein Thrombosis After Complex Posterior Spine Surgery: Does Staged Surgery Make a Difference? Spine Deform 2018;6:141-7. [Crossref] [PubMed]