
Developments in the treatment of Chiari type 1 malformations over the past decade
Introduction
Named after Austrian pathologist Hans Chiari, the Chiari malformations (CM) are a variety of structural deformities of the lower skull and cerebellum that range in severity and incidence (1). The most common and least serious variation of the Chiari syndrome is type 1. Chiari malformations type 1 (CM-1) patients aged 15 years and younger experience a 5 mm or greater displacement of the cerebellar tonsils below the foramen magnum, while patients over 15 years of age experience a 6 mm or greater displacement (2-5). These displacements typically give rise to brainstem and spinal cord compression and disrupted flow of cerebrospinal fluid (CSF) and are also thought to lead to reduced volume of the posterior fossa (3,6,7). The etiology of CM remains uncertain; however, several theories have been put forth. For instance, it has been suggested that interrupted cranial development, especially in the hindbrain, results in these structural deformities (2,4,8,9). Another theory supposes that irregular circulation of CSF alters intracranial pressure and causes CM (2,10). The pressure on the brainstem and cerebellum, caused by fetal hydrocephalus, has also been suggested as a potential cause (4). There is no consensus regarding the pathogenesis of the Chiari syndrome, as its etiology is still being researched.
While it is known that CM-1 is the most common variety of CM, the incidence of this medical condition is still relatively difficult to estimate. Studies on this matter typically report incidences of CM-1 that range from 0.56–1.00% (3,5,6,11,12), making CM-1 a fairly uncommon condition. Symptoms typically do not present until the third decade of life (12), and headaches are the most commonly-reported symptom amongst symptomatic CM-1 patients (2,6,11). However, in many cases, CM-1 patients experience no symptoms at all and find any sort of therapeutic intervention unnecessary (12).
For those CM-1 patients who do experience symptoms, there are both surgical and non-surgical treatment options. Initially, many patients undergo physical therapy or therapeutic injections to ease the burden of headaches (5). However, if symptoms are severe or persistent, there are treatment paths that include surgical intervention. The most common surgical treatment for CM-1 patients is a posterior fossa decompression surgery with or without duraplasty (2,4,13). There is currently no consensus on whether or not a duraplasty should be included in the treatment of CM-1 (8,11), although one recent study showed that the inclusion of a duraplasty is inconsequential and only raises the chances of complication (4). In some cases, however, different surgeries that target the perceived cause of CM-1 may be preferred (2). For instance, surgeons who believe disrupted CSF dynamics are the primary cause of CM-1 may seek to restore CSF flow through different types of shunt procedures (2,4,14).
To date, completed studies on the Chiari syndrome have attempted to recommend an optimal treatment path for patients by studying the efficacy of various surgeries. These studies, however, primarily consider short-term postoperative conditions and fail to examine the preoperative and intraoperative conditions of CM-1 patients. Long-term clinical outcomes of CM-1 patients are also underreported and subsequently understudied (11), and an evaluation of surgical CM-1 patients reveals that rates of symptom reoccurrence are as high as 35% (13). The prevalence of symptom reoccurrence, paired with understudied aspects of CM-1 cases, illustrates a clear need for more expansive research on the intricacies of CM-1. The present study evaluates over 5,000 patients on the larger, perioperative scale, which not only enhances our understanding of longer-term trends of CM-1 cases, but also begins to fill the scientific community’s need for more extensive and larger-scale analyses of CM-1 patients.
Therefore, the aim of this study is to quantify trends in CM-1 diagnoses and treatments using the paediatric-specific Kids’ Inpatient Database (KID) to assess fluctuations in diagnoses, treatment options, and outcomes of patients with CM-1.
Methods
Data source
The Kids’ Inpatient Database (KID) is the United States’ most expansive publicly-available all-payer pediatric (age <21 at admission) inpatient medical database. Created by the Agency for Healthcare Research and Quality’s Healthcare Cost and Utilization Project (HCUP), the KID includes information regarding complicated and uncomplicated births, as well as other inpatient procedures from community, non-rehabilitation hospitals. The KID database contains 107 data elements, using International Classification of Disease, Ninth Revision, Clinical Modification (ICD-9-CM) format to code all diagnoses and procedures until 2015. With over 3 million hospital stays per 3-year database, the KID facilitates analyses of diagnostic and therapeutic incidences using HCUP-provided trend weights (15,16). A detailed synopsis of the KID can be found at (https://www.hcup-us.ahrq.gov/kidoverview.jsp).
Patient sample
Patients in the KID database with a diagnosis of CM-1 were found using the ICD-9 code 348.4. A sub-analysis compared CM-1 malformation patients who had undergone a spinal decompression procedure (ICD-9 code 03.09) to CM-1 patients without spinal decompression performed.
Statistical analysis
IBM SPSS Statistics version 23.0 (IBM Corp., Armonk, NY, USA) was used to perform all descriptive and comparative statistics. We quantified trends of CM-1 treatments and concurrent diagnoses in the United States. Trends in demographics, procedures, and complications were analyzed using analysis of variance (ANOVA). Differences between CM-1 patients who underwent spinal decompression surgery and CM-1 patients who had not were assessed using Chi-squared and t-tests that quantified correlations among demographics, CM-1 treatments, concurrent diagnoses, complications, and secondary diagnoses. P values that were less than 0.05 were considered statistically significant.
Results
CM-1 patient demographics and comorbidities
Five thousand four hundred and thirty-eight CM-1 patients were identified in the KID from 2003–2012. The prevalence of CM-1 diagnoses has increased from 2003 to 2012, from 45 to 96 per 100,000 (P<0.001). Fifty-four point six percent of these patients were female, and the average age of these CM-1 patients was 10.51±5.71 years. Overall, 10.9% patients accounted for infantile CM-1 cases, while 36.1% of patients and 47.0% of patients represented juvenile and adolescent CM-1 cases, respectively. Fifty-nine percent of all CM-1 primary patients were white. The average length of stay (LOS) for all CM-1 patients was 4.02±7.78 days, and the average Charlson comorbidity index (CCI) for all patients was 0.213±0.519. The breakdown of demographic results based on CM-1 patients who underwent spinal decompressions and those who did not can be found in Table 1.

Full table
As a reference, the KID database as a whole contains 12,718,381. Three hundred and eighty-one patients with an average age of 6.88 years (range, 0–20 years) and is 53.8% female. The most common diagnoses in the entire KID database are pneumonia, respiratory syncytial virus-induced bronchitis, asthma, and appendicitis. One point seven percent of patients in the KID database have a diagnosis of depression and 0.8% have a diagnosis of anxiety.
From 2003–2012, the average age of all CM-1 patients steadily increased (2003: 10.02±5.57 vs. 2012: 10.76±5.71, P=0.004) (Table 2). The average CCI of all surgical patients also increased over time, from 0.16±0.46 in 2003 to 0.25±0.57 in 2012 (P<0.001) (Table 2). Over time, CM-1 patients, experienced a shorter average LOS, as the average LOS in 2003 was 4.12±2.84 days and in 2012 was 3.80±4.22 days (P=0.016) (Table 2).
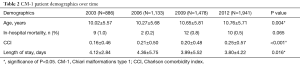
Full table
Concurrent diagnoses in CM-1 patients
Overall, 53.9% of all CM-1 patients were diagnosed with a concurrent condition. The most common concurrent diagnoses of all CM-1 patients were syringomyelia (23.8%), scoliosis (11.5%), hydrocephalus (5.9%), and various functional digestive disorders (5.5%). The most common secondary diagnoses were syringomyelia/syringobulbia (18.7%), asthma (3.4%), obstructive hydrocephalus (3.3%), and idiopathic scoliosis (2.9%) (Table 3). Sixty-three point seven percent of patients who underwent spinal decompressions were diagnosed with a concurrent condition, while only 44.46% of patients who did not have a spinal decompression had concurrent diagnoses. The incidences of most concurrent conditions were largely the same for the two groups (Table 3). However, both scoliosis (12.7% vs. 18.2%, P=0.024) and syringomyelia (27.6% vs. 32.3%, P<0.001) were more common in patients who underwent spinal decompression surgeries (Table 3).
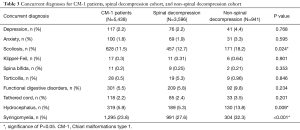
Full table
CM-1 surgical treatment
Overall, 83.4% of all CM-1 patients in the KID database underwent surgical procedures (Table 4). CM-1 surgical treatments included the following procedures: cranial decompressions, craniectomies, meninges repairs, cranial nerve decompression, spinal decompressions, fusion surgeries, other brain lesion excisions, and other spinal cord operations (Table 4). Patients were grouped on the basis of undergoing a spinal decompression: 3,569 underwent a spinal decompression and 941 underwent other surgical procedures (Table 4). One hundred percent of patients who did not undergo spinal decompressions underwent cranial decompressions, while 13% of patients who had undergone spinal decompressions underwent cranial decompressions (P<0.001) (Table 4).
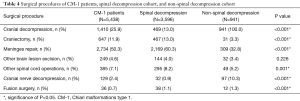
Full table
The percentage of all CM-1 patients who underwent any sort of surgery relating to their primary conditions fluctuated over time, but ultimately increased by approximately 4% (2003: 81.3% vs. 2012: 85.0%, P=0.074) (Table 5). There were notable changes in the rates of simple fusions, cranial decompressions, and spinal decompressions/laminectomies. Simple fusion rates (2003: 0.2% vs. 2012: 1.5%, P=0.021) and spinal decompression/laminectomy rates (2003: 60.8% vs. 2012: 69.0%, P<0.001) increased over time, while cranial decompression rates decreased over time (2003: 33.6% vs. 2012: 23.0%, P<0.001) (Table 5). Craniectomy rates fluctuated over the nine-year span, but the 2012 rate was ultimately similar to the 2003 rate (2003: 11.6% vs. 2006: 9.5% vs. 2009: 14.9% vs. 2012: 11.1%, P<0.001) (Table 5).

Full table
Complications of CM-1 patients
Overall, 4.8% of CM-1 patients experienced complications during their treatments for CM-1 (Table 6). The most common complications were nervous system complications (1.4%), dysphagia (0.8%), respiratory and digestive complications (0.5%), and acute respiratory distress syndrome (ARDS) (0.5%) (Table 6). The prevalence of complications was slightly higher in patients who underwent procedures other than spinal decompressions (spinal decompression: 4.7% vs. non-spinal decompression: 5.0%, P=0.547) (Table 6). Patients who did not undergo spinal decompressions experienced infection more frequently (spinal decompression: 0.1% vs. non-spinal decompression: 0.3%, P=0.021) (Table 6). The average in-hospital mortality rate across all CM-1 cases in the KID database was 0.61%, or 33 patients (Table 2). Patients who had previously undergone spinal decompressions experienced a higher mortality rate when compared to the non-spinal decompression group (spinal decompression: 0.8% vs. non-spinal decompression: 0.3%, P<0.001).
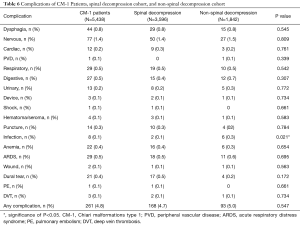
Full table
In looking at complication trends, we found that, for all CM-1 patients, complication rates decreased from 2003–2012 (2003: 5.3% vs. 2012: 2.6%, P<0.001) (Table 7). Rates of digestive complications (2003: 0.9% vs. 2012: 0%, P=0.001), dysphagia (2003: 1.5% vs. 2012: 0.2%, P<0.001), and ARDS (2003: 0.5% vs. 2012: 0%, P<0.001) decreased over time, but dural tear rates steadily increased from 2003 to 2012 (2003/2006: 0% vs. 2009: 0.4% vs. 2012: 0.7%, P=0.003) (Table 7). The mortality rate for all CM-1 patients also increased slightly from 2003 to 2012, but this was considered statistically insignificant (2003: 0.1% vs. 0.2%, P=0.582) (Table 7).
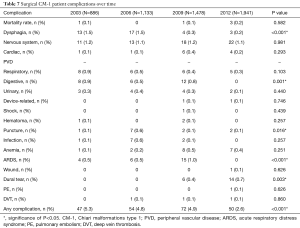
Full table
The overall complication rate for patients who underwent spinal decompressions decreased from 2003 to 2012 (2003: 6.9% vs. 2012: 2.8%, P<0.001) (Table 8); the overall complication rate for the non-spinal decompression cohort also decreased over time (2003: 5.6% vs. 2012: 3.9%, P=0.246), but this trend was considered statistically insignificant (Table 9). In 2009, there was a spike in the complication rates of ARDS for both cohorts: spinal decompression patients (2003: 0.7% vs. 2006: 0.6% vs. 2009: 1.0% vs. 2012: 0%, P=0.006) (Table 8) and non-spinal decompression patients (2003: 0% vs. 2006: 0.9% vs. 2009: 2.1% vs. 2012: 0%, P=0.019) (Table 9). For patients who had undergone spinal decompressions, there was a statistically significant decrease in the occurrence of digestive complications (2003: 1.1% vs. 2012: 0%, P=0.001) (Table 8); non-spinal decompression patients also saw a decreased rate of digestive complications (2003: 1.1% vs. 2012: 0%, P=0.081) (Table 9). Dysphagia rates decreased in the spinal decompression group (2003: 2.0% vs. 2012: 0.2%, P<0.001) (Table 8) and also in the non-spinal decompression cohort (2003: 1.1% vs. 2012: 0%, P=0.147), but the latter was considered statistically insignificant (Table 9). Dural tear rates for the spinal decompression cohort increased from 0% in 2003 to 0.9% in 2012 (P=0.011) (Table 8); dural tears also became increasingly common in the non-spinal decompression group (2003: 0% vs. 2012: 0.6%, P=0.496) (Table 9). From 2003 to 2012, the mortality rate for both groups remained nearly constant for the spinal decompression cohort (2003: 0.2% vs. 2006: 0% vs. 2009: 0.1% vs. 2012: 0.2%, P=0.595) (Table 8) and completely constant for the non-spinal decompression cohort (2003: 0% vs. 2006: 0% vs. 2009: 0% vs. 2012: 0%) (Table 9).
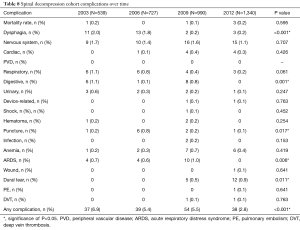
Full table
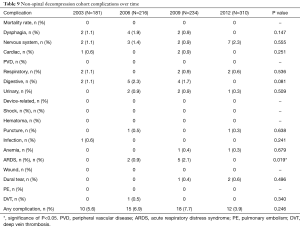
Full table
Discussion
CM-1 can negatively impact quality of life and induce problematic symptomatic responses that warrant surgical intervention. Despite this, the scientific community lacks a complete understanding of how a CM-1 patient’s pre-operative and intra-operative conditions correlate to post-operative outcomes. This study examined trends between patient outcomes and peri-operative conditions for 5,438 CM-1 pediatric patients using the KID. We found that the prevalence of CM-1 cases has approximately doubled from 2003–2012 (45 to 96 per 100,000, P<0.001). This increase in incidence is consistent with the recent findings from other studies (17), and researchers are speculating that these increased diagnoses could be a result of progress in magnetic resonance imaging, the primary diagnostic tool for the Chiari syndrome (3,6).
Preoperative trends
Examination of the results related to preoperative data yields interesting conclusions about the evolving demographics of primary CM-1 patients. The present study showed that not only have the average age and CCI score of surgical CM-1 patients increased over time, but the average age of CCI score of all CM-1 patients (including those not undergoing surgery) also increased from 2003 to 2012. Despite this higher rate of comorbidities, CM-1 patients were staying for shorter periods of time in the hospital, which could potentially be the result of improved in-hospital care. The demographic variables measured in this study (CCI, LOS, age, race, and gender) had minimal association with whether or not patients underwent a spinal decompression. However, this study did show that patients in the spinal decompression cohort more frequently had concurrent diagnoses, especially scoliosis and syringomyelia.
Surgical trends
This study also revealed that overall surgery rates of CM-1 patients has increased by 4% from 2003 to 2012, which is a trend that mirrors the increase in CM-1 diagnoses from 2003 to 2012. These findings are consistent with those of other studies, such as the recent work of Wilkinson et al. (17). Based on the findings of the present study, the increased rates of fusions and spinal decompression procedures have influenced the overall surgery rate most notably despite the decreased rates of cranial decompressions.
Postoperative trends
Despite the net increase in surgeries for CM-1 patients, there was a decrease in complication rates over time for the same cohort. This finding was unexpected and could also be linked to improved in-hospital conditions. However, from 2003 to 2012, there was an overall increase in the dural tear rate. Specifically, dural tear rates were more common in patients who had undergone spinal decompressions. When patients who had undergone spinal decompressions were compared more extensively to those who had not, we found that the complication rates between both cohorts were similar—with the non-spinal decompression group experiencing complications slightly more often, but that the non-spinal decompression cohort had a statistically significant higher mortality rate.
Limitations
We understand that there are several limitations in our study, all of which are primarily a result of the restrictions of the KID. For instance, the KID only includes inpatient data, which limits our access to information that could indicate long-term outcomes, such as radiographic images or other clinical data. Access to longer-term patient information may have influenced our analysis of complications that occurred after patient discharge. Furthermore, the KID does not include any sort of radiographic imaging, which potentially limits our understanding of the severity of certain cases. Similarly, the KID does not have the granularity to convey nuances of certain cases and details of procedures, such as surgical instruments used. In spite of these limitations, this study is the only of its kind that evaluates the trends of CM-1 cases on a perioperative scale from 2003 to 2012.
Conclusions
CM-1 diagnoses have increased from 2003 to 2012. This study showed that the average age and CCI of CM-1 patients entered into the KID have increased from 2003 to 2012 and that the average LOS has decreased during that time. Rates of surgical intervention of any kind for the treatment of CM-1 have also increased during this time. However, cranial decompression rates have decreased, as the rates of fusions and spinal decompressions have increased. Complication rates have decreased overall for all primary CM-1 patients, but dural tears have become increasingly common in the KID database from 2003 to 2012.
Acknowledgements
None.
Footnote
Conflicts of Interest: Dr. PG Passias reports personal consulting fees for Spinewave, Zimmer Biomet, and Medicrea, grants to the institution from the Cervical Spine Research Society. The other authors have no conflicts of interest to declare.
References
- Loukas M, Noordeh N, Shoja MM, et al. Hans Chiari (1851-1916). Childs Nerv Syst 2008;24:407-9. [Crossref] [PubMed]
- Akar E, Kara S, Akdemir H, et al. 3D structural complexity analysis of cerebellum in Chiari malformation type I. Med Biol Eng Comput 2017;55:2169-82. [Crossref] [PubMed]
- Raza-Knight S, Mankad K, Prabhakar P, et al. Headache outcomes in children undergoing foramen magnum decompression for Chiari I malformation. Arch Dis Child 2017;102:238-43. [Crossref] [PubMed]
- Chen J, Li Y, Wang T, et al. Comparison of posterior fossa decompression with and without duraplasty for the surgical treatment of Chiari malformation type I in adult patients: A retrospective analysis of 103 patients. Medicine (Baltimore) 2017;96:e5945. [Crossref] [PubMed]
- McVige JW, Leonardo J. Neuroimaging and the clinical manifestations of Chiari Malformation Type I (CMI). Curr Pain Headache Rep 2015;19:18. [Crossref] [PubMed]
- Speer MC, Enterline DS, Mehltretter L, et al. Review Article: Chiari Type I Malformation with or Without Syringomyelia: Prevalence and Genetics. J Genet Couns 2003;12:297-311. [Crossref] [PubMed]
- Badie B, Mendoza D, Batzdorf U, et al. Posterior fossa volume and response to suboccipital decompression in patients with Chiari I malformation. Neurosurgery 1995;37:214-8. [Crossref] [PubMed]
- Shoja MM, Johal J, Oakes WJ, et al. Embryology and Pathophysiology of the Chiari I and II Malformations: A Comprehensive Review. Clin Anat 2018;31:202-15. [Crossref] [PubMed]
- Milhorat TH, Nishikawa M, Kula RW, et al. Mechanisms of cerebellar tonsil herniation in patients with Chiari malformations as guide to clinical management. Acta Neurochir (Wien) 2010;152:1117-27. [Crossref] [PubMed]
- Cai C, Oakes WJ. Hindbrain herniation syndromes: The chiari malformations (I and II). Semin Pediatr Neurol 1997;4:179-91. [Crossref] [PubMed]
- Kalb S, Perez-Orribo L, Mahan M, et al. Evaluation of operative procedures for symptomatic outcome after decompression surgery for Chiari type I malformation. J Clin Neurosci 2012;19:1268-72. [Crossref] [PubMed]
- Meadows J, Kraut M, Guarnieri M, et al. Asymptomatic Chiari Type I malformations identified on magnetic resonance imaging. J Neurosurg 2000;92:920-6. [Crossref] [PubMed]
- McGirt MJ, Attenello FJ, Atiba A, et al. Symptom recurrence after suboccipital decompression for pediatric Chiari I malformation: Analysis of 256 consecutive cases. Childs Nerv Syst 2008;24:1333-9. [Crossref] [PubMed]
- Brugières P, Idy-Peretti I, Iffenecker C, et al. CSF flow measurement in syringomyelia. AJNR Am J Neuroradiol 2000;21:1785-92. [PubMed]
- Healthcare cost and utlization project (HCUP). Introduction to the HCUP National Inpatient Sample (NIS) 2013. Available online: https://www.hcup-us.ahrq.gov/db/nation/nis/NIS_Introduction_2013.jsp
- Taylor CA, Greenspan AI, Xu L, et al. Comparability of national estimates for traumatic brain injury-related medical encounters. J Head Trauma Rehabil 2015;30:150-9. [Crossref] [PubMed]
- Wilkinson DA, Johnson K, Garton HJ, et al. Trends in surgical treatment of Chiari malformation Type I in the United States. J Neurosurg Pediatr 2017;19:208-16. [Crossref] [PubMed]


