
An awake, minimally-invasive, fully-endoscopic surgical technique for treating lumbar radiculopathy secondary to heterotopic foraminal bone formation after a minimally invasive transforaminal lumbar interbody fusion with BMP: technical note
Introduction
Several advantages of minimally invasive transforaminal lumbar interbody fusion (MIS-TLIF) over the open version of the procedure have been described: less blood loss, less pain, shorter hospitalization, and earlier rehabilitation (1). One disadvantage of the minimally invasive procedure is the challenge to the surgeon’s ability to perform adequate disc preparation while constrained to working down a small tubular retractor. The use of recombinant human bone morphogenetic protein (rhBMP-2) is a reliable adjunct used to augment the fusion process but several possible complications have been suggested to be associated with its use: radiculitis, cancer, osteolysis, retrograde ejaculation, and heterotopic bone formation (2-11). The procedure presented here is an awake, endoscopic foraminal decompression that utilizes an oblique trajectory through Kambin’s triangle, avoids scar tissue, and does not risk exposing the patient’s hardware.
Case presentation
History and presentation
A 42-year-old male underwent a lumbar 4–sacral 1 MIS-TLIF by another surgeon. He did well in the initial postoperative period, but he began to note right leg pain and numbness in an L5 dermatomal pattern. The pain continued for 2 years despite interventional pain management, and he began to note left foot dorsiflexion weakness. An electromyography (EMG) study showed a right L5 radiculopathy and a computed tomography (CT) lumbar spine demonstrated excessive bone growth in the right L4–5 neural foramen. The patient underwent an awake, endoscopic foraminotomy procedure utilizing a blunt tipped manual shaver drill system. The patient’s radicular symptoms improved immediately, and he remained asymptomatic at the 1 year follow up.
Operative procedure
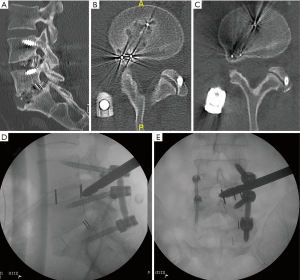
For the transforaminal endoscopic procedure to perform the foraminotomy at L4–5 on the right, the patient was positioned prone on the Wilson frame and the procedure was done under local analgesia with intravenous sedation; the level of anesthetic was titrated so the patient was able to communicate with the surgeon throughout the procedure. The Joimax® (Germany) TESSYS® endoscopic system was used for the procedure. Percutaneous entry was established entering through the skin 11 cm lateral to the midline. Using intermittent fluoroscopic guidance, alternating between lateral and anterior-posterior (AP) view, a 25 cm 18 gauge needle was advanced and placed at the superior endplate of L5 through Kambin’s triangle, between the exiting and traversing nerves. An AP fluoroscopic view was used to assure that at the point the needle touched the superior endplate of L5 the needle was not past the medial wall of the L5 pedicle. Sequential reamers were used to enlarge the neural foramen by removing the ventral aspect of the superior facet. Sequential side shaver manual drills were then used to safely expand the foramen. Figure 1 depicts the largest side shaver drill demonstrated in the lateral (Figure 1D) and AP fluoroscopic (Figure 1E) views. A very aggressive trajectory of the side shaver drill was possible because the drill’s tip is blunt and the instrument can effectively protect the traversing nerve root and thecal sac by pushing them forward as the sides of the drill remove bone. The technique used, while rotating the drill manually, was to lift the drill to shave the roof of the foramen (the superior articulating process of L5), and lower the drill to shave the bony overgrowth. After the percutaneous drilling was performed, the drill was removed, and the working channel endoscope was placed in the foramen—endoscopic graspers were used to remove remaining fragments of bone and ligamentum flavum until it was evident that the exiting L4 and traversing L5 nerve roots were no longer compressed. The patient was asked prior to terminating the procedure the status of his radicular symptoms. He confirmed the relief of his radicular symptoms and the working channel and scope were removed, pressure was held on the 5 mm incision for 5 minutes, and the wound was closed with a single interrupted suture.
Postoperative course
The postoperative course was uneventful, and the patient’s pain improved immediately after surgery. One year after his endoscopic procedure, the patient had no clinical symptoms related to the heterotopic bone formation. Figure 1B and 1C demonstrates on axial CT the completely stenotic right L4–5 foramen (Figure 1B) and the more patent (albeit there remains significant heterotopic bone) foramen on the 6-month postoperative CT (Figure 1C).
Discussion
The use of rhBMP-2 has helped spine surgeons enhance fusions, reduce the morbidity involved in harvesting autograft, and allow surgeons to attempt more and more minimally invasive spine fusions (12,13). The use of biologics to enhance fusion, however, does not come without complications or expense. The complication treated here, heterotopic bone formation, required an additional surgery to treat. Another surgical technique, also minimally invasive, has been presented in the literature and describes resecting the heterotopic bone formation through a tubular retractor using a posterior micro endoscopic procedure (3). The advantage of this type of approach is a more perpendicular approach to the heterotopic bone which allows direct “drilling-down” of the compressive pathology. The disadvantages of this posterior approach are that in the setting of a previous posterior surgery and possible previous violation of the dura, the possible risks for neural injury (because of difficult scar dissection) and spinal fluid leak are probably greater. The approach suggested here avoids previous scar and does not disrupt previous fusion mass.
There are two significant short-comings to the technique presented here. First, the manual side shaver drill is used with only fluoroscopy for visualization: it is a percutaneous decompression with subsequent endoscopic visualization. The surgeon is guided by the location of the tip of the drill on AP and lateral fluoroscopy and the patient’s symptomatic feedback. Second, the post-op CT shows residual heterotopic foraminal bone. The right L4–5 foramen is less stenotic and the patient’s symptoms are improved, but additional decompression of the heterotopic bone could have been performed under direct visualization with an endoscopic drill.
The author and colleagues have previously published other endoscopic surgical techniques as “rescue” procedures for the complications seen after other spine surgical procedures: kyphoplasty (14), instrumented spine fusion (15), TLIF (16), artificial disc (17), adjacent segment disease (18) and thoracic spine tumor recurrence (19). Patients who have already undergone significant spine surgical procedures are often hesitant to pursue additional surgeries and often travel great distances seeking an awake, endoscopic solution to their problem.
Conclusions
Minimally invasive spine surgery offers many benefits that are attractive to patients: shorter recovery times, small incisions, and less pain. Hence, minimally invasive spine surgery procedures are being performed more and more. With the increase in MIS procedures being performed, surgical practitioners are put in a situation where they have to get used to treating the unique complications that now arise from these procedures. Encouraging, though, is the fact that with the increase in minimally invasive techniques in the spine surgery armamentarium, there are more minimally invasive techniques to, somewhat ironically, treat the complications involved in MIS spine surgery.
Acknowledgements
None.
Footnote
Conflicts of Interest: The author has no conflicts of interest to declare.
Informed Consent: Informed consent has been obtained from the subject of this study.
References
- Seng C, Siddiqui MA, Wong KP, et al. Five-year outcomes of minimally invasive versus open transforaminal lumbar interbody fusion: a matched-pair comparison study. Spine (Phila Pa 1976) 2013;38:2049-55. [Crossref] [PubMed]
- Ahn J, Jorgensen AY, Bohl DD, et al. Neuroforaminal Bone Growth Following Minimally Invasive Transforaminal Lumbar Interbody Fusion With BMP: A Computed Tomographic Analysis. Clin Spine Surg 2017;30:E754-8. [Crossref] [PubMed]
- Ahn J, Tabaraee E, Singh K. BMP-2-induced Neuroforaminal Bone Growth in the Setting of a Minimally Invasive Transforaminal Lumbar Interbody Fusion. J Spinal Disord Tech 2015;28:186-8. [Crossref] [PubMed]
- Carragee EJ, Chu G, Rohatgi R, et al. Cancer risk after use of recombinant bone morphogenetic protein-2 for spinal arthrodesis. J Bone Joint Surg Am 2013;95:1537-45. [Crossref] [PubMed]
- Carragee EJ, Hurwitz EL, Weiner BK. A critical review of recombinant human bone morphogenetic protein-2 trials in spinal surgery: emerging safety concerns and lessons learned. Spine J 2011;11:471-91. [Crossref] [PubMed]
- Chen NF, Smith ZA, Stiner E, et al. Symptomatic ectopic bone formation after off-label use of recombinant human bone morphogenetic protein-2 in transforaminal lumbar interbody fusion. J Neurosurg Spine 2010;12:40-6. [Crossref] [PubMed]
- Comer GC, Smith MW, Hurwitz EL, et al. Retrograde ejaculation after anterior lumbar interbody fusion with and without bone morphogenetic protein-2 augmentation: a 10-year cohort controlled study. Spine J 2012;12:881-90. [Crossref] [PubMed]
- Joseph V, Rampersaud YR. Heterotopic bone formation with the use of rhBMP2 in posterior minimal access interbody fusion: a CT analysis. Spine (Phila Pa 1976) 2007;32:2885-90. [Crossref] [PubMed]
- Lindley EM, McBeth ZL, Henry SE, et al. Retrograde ejaculation after anterior lumbar spine surgery. Spine (Phila Pa 1976) 2012;37:1785-9. [Crossref] [PubMed]
- Singh K, Ahmadinia K, Park DK, et al. Complications of spinal fusion with utilization of bone morphogenetic protein: a systematic review of the literature. Spine (Phila Pa 1976) 2014;39:91-101. [Crossref] [PubMed]
- Villavicencio AT, Burneikiene S. RhBMP-2-induced radiculitis in patients undergoing transforaminal lumbar interbody fusion: relationship to dose. Spine J 2016;16:1208-13. [Crossref] [PubMed]
- Lad SP, Nathan JK, Boakye M. Trends in the use of bone morphogenetic protein as a substitute to autologous iliac crest bone grafting for spinal fusion procedures in the United States. Spine (Phila Pa 1976) 2011;36:E274-81. [Crossref] [PubMed]
- Singh K, Nandyala SV, Marquez-Lara A, et al. Epidemiological trends in the utilization of bone morphogenetic protein in spinal fusions from 2002 to 2011. Spine (Phila Pa 1976) 2014;39:491-6. [Crossref] [PubMed]
- Wagner R, Telfeian AE, Iprenburg M, et al. Transforaminal Endoscopic Solution to a Kyphoplasty Complication: Technical Note. World Neurosurg 2016;91:195-8. [Crossref] [PubMed]
- Telfeian AE, Jasper GP, Francisco GM. Transforaminal endoscopic treatment of lumbar radiculopathy after instrumented lumbar spine fusion. Pain Physician 2015;18:179-84. [PubMed]
- Telfeian AE. Transforaminal Endoscopic Solution to Disc Reherniation Post-mini-TLIF: Technical Note. Clin Neurol Neurosurg 2015;131:69-71. [Crossref] [PubMed]
- Wagner R. Iprenburg, Telfeian AE. Transforaminal endoscopic decompression of a postoperative dislocated bone fragment after a 2-level lumbar total disc replacement: case report. Neurosurg Focus 2016;40:E8. [Crossref] [PubMed]
- Telfeian AE. Transforaminal Endoscopic Surgery for Adjacent Segment Disease After Lumbar Fusion. World Neurosurg 2017;97:231-5. [Crossref] [PubMed]
- Telfeian AE, Choi DB, Aghion DM. Transforaminal endoscopic surgery under local analgesia for ventral epidural thoracic spinal tumor: Case report. Clin Neurol Neurosurg 2015;134:1-3. [Crossref] [PubMed]


