Minimally invasive surgery in adult degenerative scoliosis: a systematic review and meta-analysis of decompression, anterior/lateral and posterior lumbar approaches
Introduction
Open surgery has traditionally been employed for adult lumbar degenerative deformities, often with multi-level decompression and fusion to stabilize the columns and reduce neural compression. However, there has been a surge in the use of minimally invasive approaches for the treatment of multi-level pathology including adult degenerative scoliosis. Minimally invasive approaches were introduced to address approach-related morbidity associated with open spine surgery, with increasing applications to more complex patient pathologies (1). Less invasive surgery has the potential to minimize blood loss, reduce surgical trauma and stress to muscles and paraspinal structures, reduce analgesic use and reduce hospital stay. Minimally invasive approaches for degenerative scoliosis reported in the literature includes decompression only, lateral minimally invasive thoracolumbar instrumentation, minimally invasive posterior, transforaminal and anterior fusion approaches.
Though minimally invasive fusion has been associated with good initial results, most series discussing minimally invasive spinal (MIS) fusion have been in the presence of short-segment fusion (2-6). However, direct comparison of the safety and complication profiles of different minimally invasive surgical approaches for adult degenerative scoliosis remain scarce. Given that lateral LIF (LLIF) is a transpsoas approach, it is expected that there may be higher rates of motor and sensory deficits. Decompression alone approaches may be expected to yield higher revision rates with lower rates of satisfaction. Given the limited comparative evidence on this topic, this study aims to use meta-analytical techniques to compare the different minimally invasive surgical approaches for adult degenerative scoliosis with respect to clinical outcomes, changes in radiographic measurements including Cobb angle and lumbar lordosis, and complication profiles.
Methods
Literature search strategy
The Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines were followed for the present systematic review. Electronic searches were performed using Ovid Medline, PubMed, Cochrane Central Register of Controlled Trials (CCTR), Cochrane Database of Systematic Reviews (CDSR), ACP Journal Club, and Database of Abstracts of Review of Effectiveness (DARE), from their dates of inception to September 2015. To achieve maximum sensitivity of the search strategy, we combined the terms “minimally invasive” or “fusion” or “decompression” and “degenerative scoliosis” and “adult” which were searched as text words and exploded as MeSH headings where possible. Two authors performed the search independently, and any discrepancies were resolved by discussion. The reference lists of all retrieved articles were reviewed for further identification of potentially relevant studies, assessed using the inclusion and exclusion criteria. Expert academic spinal surgeons were consulted as to whether they knew of any unpublished data (7).
Selection criteria
Eligible studies for the present systematic review and meta-analysis included those in which patient cohorts underwent minimally invasive surgery for adult degenerative scoliosis. When institutions published duplicate studies with accumulating numbers of patients or increased lengths of follow-up, only the most complete reports were included for quantitative assessment. All publications were limited to those involving human subjects and in the English language. Abstracts, case reports, conference presentations, editorials, reviews and expert opinions were excluded.
Data extraction and critical appraisal
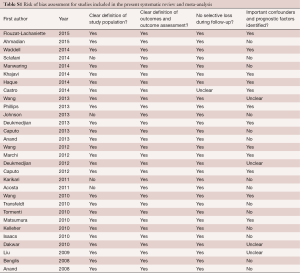
All data including baseline characteristics, operational parameters, and safety and efficacy outcomes were extracted from article texts, tables and figures. The primary outcome was fusion rate at follow-up as well as change in Cobb angle. Other outcomes extracted included: change in visual analogue scale (VAS) back pain score, change in Oswestry disability index (ODI) score, change in lumbar lordosis angle, and complication rates. Two investigators independently reviewed each retrieved article. Discrepancies between the two reviewers were resolved by discussion and consensus. The quality of studies was assessed using criteria recommended by the National Health Service Centre for Reviews and Dissemination case series quality assessment criteria (University of York, Heslington, United Kingdom). The final results were reviewed by the senior investigators.
Statistical analysis
Data are presented as mean ± standard deviation. For weighted pooled means, a meta-analysis of proportions was conducted. Firstly, to establish variance of raw proportions, a transformation was applied. To incorporate heterogeneity (anticipated among the included studies), transformed proportions were combined using DerSimonian-Laird random effects models. Finally the pooled estimates were back-transformed. Heterogeneity was evaluated using Cochran Q and I2 test. Weighted means were calculated by determining the total number of events divided by total sample size.
A formal statistical comparison was performed between decompression, anterior, lateral and transforaminal fusion approaches using mixed-effects meta-regression with a fixed-effect moderator variable for interventional technique. All analyses were performed using the metafor package for R version 3.02. P values <0.05 were considered statistically significant.
Results
Search strategy
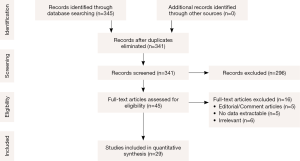
A total of 345 references were identified from the electronic databases search. After exclusion and exclusion criteria were applied, 45 references remained for full text evaluation (Figure 1). Manual reference list searches did not yield additional studies. After final application of criteria, there were 29 studies (8-20) (1,228 patients) (21-36) included for qualitative and quantitative analysis in the present study. All studies were observational studies, with mean follow-up range of 4.75 to 68.4 months (Table 1). Risk of bias assessment for each included study is summarized in Table S1.
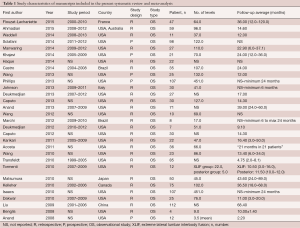
Full table
Full table
Baseline characteristics
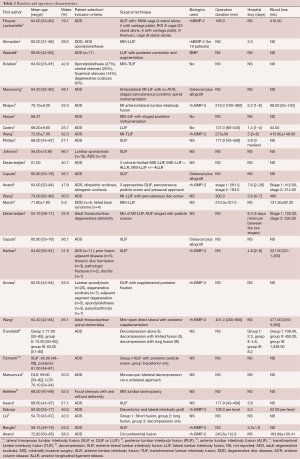
Patient age ranged from 54 to 77 years. Inclusion and exclusion criteria varied between studies. All required at minimum a diagnosis of adult degenerative lumbar scoliosis at some level with disability, and have undergone a minimally invasive lumbar fusion. The mean operative time ranged from 137 to 401 minutes. The mean hospital stay ranged from 1.4 to 7.6 days. The blood loss ranged from 54 to 480 mL. These characteristics for each study are shown in Table 2.
Full table
Functional outcomes: VAS & ODI scores
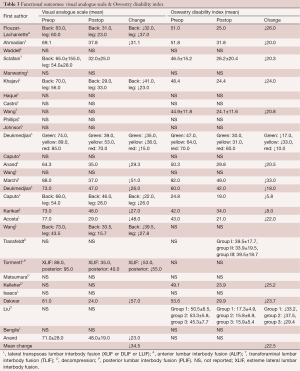
All the studies demonstrated a decrease in pain post-operation compared to pre-operation, as measured by the VAS. The mean decrease in VAS was 34.5 points. The mean pre-operative VAS ranged from 43.5 to 95. Mean post-operative VAS ranged from 15.7 to 70 points. Tormenti and colleagues (32) demonstrated the decrease in VAS pain scores was similar between the LLIF and posterior approach (53 vs. 55). In the four studies (14,19,25,36) which separately measured back and leg pain, back pain was consistently reported to be worse than the leg pain pre-operatively (range: 3 to 29.5 points difference). A greater degree of pain reduction was reported for back pain compared to leg pain (33.6 & 28.5 points, respectively) (Table 3).
Full table
In terms of disability, all studies demonstrated a decrease after the operation, as measured by the ODI (range: 8 to 37.5). The mean decrease in ODI was 22.5 points. The mean pre-operative ODI ranged from 24.8 to 82 points (Table 3).
Radiological outcomes: Cobb angle & Lumbar lordosis
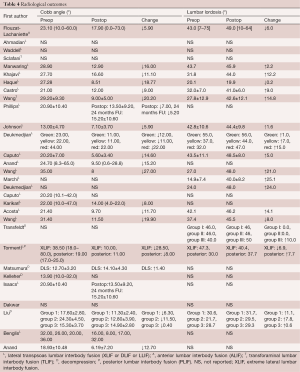
All the studies, except one, demonstrated a decrease in the Cobb angle (range: –20.2 to +1.4) post-operatively. Liu demonstrated the decrease in Cobb angle was greatest for long fusion (–11.5), followed by short fusion (–6.3), and least for decompression only (–0.4). Tormenti demonstrated the decrease in Cobb angle was greater for LLIF than the posterior approach (28.5 vs. 8). The pre-operative Cobb angle ranged from 12.7 to 38.5. The post-operative Cobb angle ranged from 5.6 to 32 (Table 4).
Full table
All studies, except four, demonstrated an increase in the lumbar lordosis angle (range: –6.9 to 25.1). Tormenti and colleagues (32) demonstrated LLIF achieved a mean decrease of 6.9°, whilst the posterior approach increased the lumbar lordosis angle by 7.7°. Transfeldt and colleagues (33) demonstrated decompression with long fusion achieved an increase of 10° for lumbar fusion, however, no change was seen for decompression alone or decompression with limited fusion.
Fusion rates
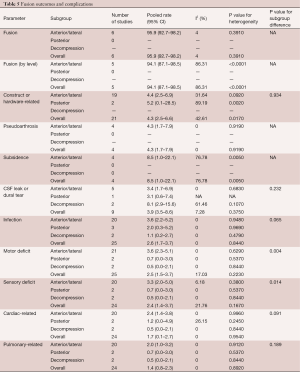
Fusion rate (by proportion of patients) was reported only in studies, which used a minimally invasive lateral or LLIF approach. The pooled fusion rate was 95.9% (95% CI: 92.7–98.2%). Fusion rate according to proportion of levels was reported for the LLIF technique in six studies. Pooled fusion rate according to per level was 94.1% (95% CI: 87.1–98.5%) (Table 5).
Full table
Construct and hardware complications
Total pooled rates for all minimally invasive approaches used was 4.3% (95% CI: 2.4–6.7%). Rate of construct or hardware complication was similar among the different surgical approaches for adult degenerative scoliosis. Pooled construct or hardware complication rate was 4.4% (95% CI: 2.5–6.9%) for LLIF and 5.2% (95% CI: 0.1–28.5%) for transforaminal LIF (TLIF).
Pseudoarthrosis
Pseudoarthrosis was reported in six included studies in this meta-analysis. The pooled pseudoarthrosis rate from the four LLIF studies and two decompression studies was 4.3% (95% CI: 1.7–7.9%).
Subsidence
Subsidence rates were also reported in four LLIF studies. There was significant heterogeneity (I2=77%, P=0.005) detected between the studies, with Castro et al. (29%), Johnson et al. (22) (3.3%), Karikari et al. (23) (4.5%), and Dakwar et al. (16) (4.0%) reporting different rates. This is likely because Castro et al. (15) had a longer follow-up period of up to 24 months, which may have captured higher rates of subsidence. The pooled subsidence rate for LLIF studies was 8.5% (95% CI: 1.0–22.1%).
Dural tears or CSF leak
The total pooled rate of dural tears and CSF leaks across the included studies was 5.8% (3.5–8.6%). In the LLIF group, pooled rates of tears and CSF leak was 5.4% (95% CI: 1.7–10.9%), compared to 3.1% (0.6–7.4%) in the TLIF group and 8.1% (2.9–15.6%) in the decompression group. No significant difference was detected among the rate of infections reported (P=0.232).
Infections
Pooled infection rates across 25 studies was 2.6% (95% CI: 1.7–3.7%). When subgrouped according to surgical approach, the anterior/lateral approach rate was 3.6% compared to minimally invasive TLIF (2.0%) and decompression (1.1%). These differences were trending towards significance (P=0.065).
Motor and sensory deficits
The overall pooled rate of motor deficit for all minimally invasive surgery for degenerative lumbar scoliosis was 2.5% (95% CI: 1.5–3.7%). From 21 anterior/lateral minimally invasive approaches, the pooled motor deficit rate was 3.6% (95% CI: 2.3–5.1%). This was significantly higher compared to minimally invasive TLIF (0.7%, 95% CI: 0–3.0%) and decompression (0.5%, 95% CI: 1.5–3.7%) (P=0.004).
The total pooled rate of sensory deficit from all minimally invasive surgical approaches used including fusion and decompression was 2.4% (95% CI: 1.4–3.7%). For the minimally invasive LLIF approach, the pooled sensory deficit rate was 3.3% (95% CI: 2.0–5.0%). Minimally invasive TLIF had a pooled sensory deficit rate of 0.7% (95% CI: 0–3.0%), whilst decompression-only resulted in pooled rate of 0.5% (95% CI: 0–2.1%). Meta-regression analysis demonstrated significant difference between the approaches used (P=0.014).
Cardiac events
The total pooled rates for all minimally invasive approaches used were 1.7% (95% CI: 0.1–2.7%). The rate of cardiac complications was similar (P=0.091) among the different surgical approaches for adult degenerative scoliosis.
Pulmonary events
Total pooled rates for all minimally invasive approaches used was 1.4% (95% CI: 0.8–2.3%). Rate of pulmonary complications was similar (P=0.189) among the different surgical approaches for adult degenerative scoliosis.
Discussion
Lumbar degenerative scoliosis is a common degenerative condition of the lumbar spine associated with considerable morbidity. Although the etiology of this condition is not clear, the most commonly implicated causes include asymmetrical degeneration of discs, osteoporosis and vertebral body compression fractures (37). Radiological features include facet hypertrophy, loss of lumbar lordosis and increasing deformity in sagittal and coronal planes. Affected patients most commonly complain of axial low back pain with or without radiculopathy, with stenotic symptoms localized to the primary lumbar curve, generally without neurologic deficit (38-40). This pain may be generated directly by the facet joints or due to nerve root impingement or traction.
The surgical treatment for symptomatic adult scoliosis remains controversial, namely due to the extensive morbidity associated with the conventional, open, surgical approaches (41-44). These open approaches have reported complication rates ranging from 28.1% to 66% with extensive operative time, hospitalization, recovery and return to normal activity (45,46). Despite the risks, these surgical interventions have shown greater benefits over non-surgical treatment in decreasing pain and disability, whilst increasing the health-related quality of life (HRQOL) (41,47). However, major open surgery is often limited by the patients’ age, medical comorbidities, as well as the considerable blood loss expected during open surgery.
MIS fusion has been increasingly used as it has been associated with decreased blood loss, decreased hospital stays, and decreased pain compared to open fusion (5). One minimally invasive approach is the decompression procedure such as decompressive laminectomy with or without foraminotomy. Fusion is also an option, which has an increasing array of surgical approaches available. The majority of surgical approaches involved anterior column support with the fusion, and posterior instrumentation. Approaches for lumbar fusion include the: lateral transpsoas interbody fusion [LLIF/DLIF/extreme lateral lumbar interbody fusion (XLIF)], anterior lumbar interbody fusion (ALIF), TLIF and posterior lumbar interbody fusion (PLIF).
Deformity correction
This review demonstrates minimally invasive surgery for adult lumbar scoliosis was able to correct for deformities, with outcomes similar to open surgery. The greatest improvement in the Cobb angle and lumbar lordosis is seen with fusion techniques compared to decompression alone. Liu and colleagues examined minimally invasive short fusion, long fusion and decompression alone. Patients with decompression alone had the lowest change in Cobb angle (decrease of 0.4°) and lumbar lordosis (increase of 0.6°), whilst long fusion had the greatest change in Cobb angle (decrease of 11.5°) and lumbar lordosis (increase of 7.8°). Wang and colleagues, who investigated 23 patients, reported one of the highest decreases in Cobb angle of 27° (pre-operative: 35° to post-operative: 8°). This study used a mini-open direct lateral approach with posterior supplementation. Tormenti and colleagues evaluated patients who underwent XLIF with posterior pedicle screw, and those with the posterior approach only. They identified the group with XLIF with the posterior pedicle screw achieved a greater decrease in Cobb angle of 28.5° compared to the posterior approach alone (decrease of 8°). However, this may be due the large Cobb angle pre-operatively for the XLIF group (38.5°) compared to the posterior approach alone (19°). Both approaches achieved a similar post-operative Cobb angle (10° & 11°). To date there is evidence that suggests minimally invasive lateral procedures are more effective in correcting coronal deformities than sagittal deformities such as regional lordosis, which is more clinically significant (48). While studies are still limited, there are newer minimally invasive techniques such as anterior column realignment (ACR), which can effectively correct sagittal deformities with potentially less surgical complications (49,50). Nevertheless, the current evidence suggests that minimally invasive fusion approaches may be associated with improved deformity correction. However, further long-term studies are required to determine the differences in deformity progression between the anterior, lateral, and posterior fusion techniques.
Clinical outcome
The results from this review demonstrated that minimally invasive surgical approaches are effective at improving the functional outcomes of degenerative scoliosis patients, with rates similar to open, conventional procedures. All the included studies demonstrated a decrease in back pain and disability post-operation compared to pre-operation, as measured by the VAS and the ODI respectively. The pooled absolute decrease in the VAS back pain score was 34.5 points (pre-operative range, 43.5–95 points & post-operative range, 15.7–70 points). The pooled absolute decrease in ODI was 22.5 points (range, 8–37.5). Tormenti et al. demonstrated the decrease in VAS pain scores was similar between XLIF and posterior approach (53 vs. 55 points). Liu and colleagues investigated 112 patients and demonstrated the average improvement in the ODI was 32.6, 26.3 and 13.5 for long segment fusion, short segment fusion and simple decompression without fusion (mean of 5.7 years follow-up). However, as seen from Table 3, there are several studies that have not reported VAS or ODI data. Therefore, it is difficult to make firm definitive conclusions. Further research is warranted to compare the clinical outcome differences between the different minimally invasive approaches.
Complications
The total pooled fusion and pseudoarthrosis rates for all minimally invasive surgery for degenerative lumbar scoliosis were 95.9% and 6.0%, respectively. Meta-regression demonstrated that pseudoarthrosis rates were similar between anterior/lateral approaches compared to decompression (4.3% vs. 7.5%, respectively) (P=0.189).
The mean overall pooled rate of motor deficit and sensory deficit was 2.5% and 2.4% respectively. Significantly higher motor deficits were seen in the anterior/lateral approach compared to the transforaminal approach and decompression alone. Similar trends were also seen for sensory deficit, which was significantly higher in the anterior/lateral subgroup. The significantly higher rate of motor deficits for the anterior/lateral can be justified by the fact that LLIF requires dissection of the psoas major, which may injure the nerves of the lumbar plexus or cause significant trauma to the psoas major. A possible explanation for the higher rates of motor deficits for ALIF may be related to the violation or retraction of great vessels, whereby undetected injury or intraoperative ischemia (51,52) may result in post-operative motor deficits. This may be further compounded by the increased operative time for ALIF compared to XLIF (53,54). Closer examination into the studies which used a more anterior corridor demonstrates the motor deficit reported in one study (25) was foot drop in 1 patient (4.8%), and in the other study (19), a persisting, complete, L5 palsy without residual compression on CT scan in 1 patient (2.1%) and acute urinary retention in 6 patients (12.8%). If the patients who developed acute urinary retention were removed, the rates of motor deficits for ALIF would be 3.45%, which would be similar to a purely lateral approach.
The total pooled rate of infections, dural tears/CSF leaks, hardware complications, cardiac and pulmonary events were 2.6%, 5.8%, 4.3%, 1.7% and 1.4%, respectively. There were no significant differences between the different minimally invasive interbody fusion techniques. The rates of complication in this review are substantially lower than open approaches, which have reported rates ranging from 28.1% to 66% (45,46).
Learning curve and comparison with open surgery
MIS fusions have been associated with steep learning curves, increased surgical times, and increased radiation exposure. However, Anand and colleagues (11) have demonstrated otherwise, where MIS approaches were technically feasible, had shorter hospital stays, able to be accomplished within very reasonable operative times, and associated with much less blood loss than open procedures (when compared with the literature). Additionally, this review illustrates the lengths of surgery, hospital stay and blood loss associated with MIS fusions for lumbar degenerative scoliosis is relatively lower than open procedures (Table 2). Furthermore, the clinical outcomes, both in terms of VAS and ODI demonstrate excellent results for minimally invasive procedures.
Limitations
Limitations of the current review include the lack of direct comparative studies between the different minimally invasive surgical approaches (1,55). This resulted in significant heterogeneity and selection bias unaccounted for. In order to minimise heterogeneity, subgroup analysis was performed based on the type of fusion and separated decompression only studies out. However, there still remains a significant level of heterogeneity regarding the techniques used by different surgeons and centres (e.g., type of posterior instrumentation, graft types, additional posterior instrumentation). Additionally, the follow-up duration was variable between studies and limited for some studies [2.2 months (11)]. This may undermine the true rate of complications in studies which have a relatively shorter follow up compared to those with a longer follow up, such as the rate of pseudoarthrosis, changes in Cobb angle and lumbar lordosis. However, the effect of this is reduced by having a majority of pooled studies into the meta-regression having more than 12 months follow-up. Poor reporting of key outcomes from the included studies also limited assessment of surgical approaches. For example, few studies reported SVA as a marker of sagittal correction, and it was difficult to compare statistically blood loss, operative time, and length of stay among the approaches. Despite these limitations, this review has several strengths such as thoroughly evaluating and assessing the functional and clinical outcome of the available literature for minimally invasive surgery for adult degenerative scoliosis.
Conclusions
Minimally invasive spine technologies may be used for the surgical treatment of lumbar degenerative scoliosis. The current review adds to the growing literature examining minimally invasive techniques in adult scoliosis, suggesting that the procedure may have acceptable complication rates, radiological outcomes and clinical outcomes. Anterior and particularly lateral approaches are likely associated with increased motor and sensory deficit compared to posterior approaches. Similar rates of hardware/constructed-related complications, CSF leak, cardiac and pulmonary complications were found among LLIF, TLIF and decompression techniques. Future studies, specifically multi-centered longitudinal, examining the adequacy of MIS is warranted to compare long-term outcomes with the traditional procedure.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Mobbs RJ, Phan K, Malham G, et al. Lumbar interbody fusion: techniques, indications and comparison of interbody fusion options including PLIF, TLIF, MI-TLIF, OLIF/ATP, LLIF and ALIF. J Spine Surg 2015;1:2-18.
- Anand N, Hamilton JF, Perri B, et al. Cantilever TLIF with structural allograft and RhBMP2 for correction and maintenance of segmental sagittal lordosis: long-term clinical, radiographic, and functional outcome. Spine (Phila Pa 1976) 2006;31:E748-53. [Crossref] [PubMed]
- Deutsch H, Musacchio MJ Jr. Minimally invasive transforaminal lumbar interbody fusion with unilateral pedicle screw fixation. Neurosurg Focus 2006;20:E10. [Crossref] [PubMed]
- Jang JS, Lee SH. Minimally invasive transforaminal lumbar interbody fusion with ipsilateral pedicle screw and contralateral facet screw fixation. J Neurosurg Spine 2005;3:218-23. [Crossref] [PubMed]
- Park Y, Ha JW. Comparison of one-level posterior lumbar interbody fusion performed with a minimally invasive approach or a traditional open approach. Spine (Phila Pa 1976) 2007;32:537-43. [Crossref] [PubMed]
- Schwender JD, Holly LT, Rouben DP, et al. Minimally invasive transforaminal lumbar interbody fusion (TLIF): technical feasibility and initial results. J Spinal Disord Tech 2005;18 Suppl:S1-6. [Crossref] [PubMed]
- Phan K, Mobbs RJ. Systematic reviews and meta-analyses in spine surgery, neurosurgery and orthopedics: guidelines for the surgeon scientist. J Spine Surg 2015;1:19-27.
- Acosta FL, Liu J, Slimack N, et al. Changes in coronal and sagittal plane alignment following minimally invasive direct lateral interbody fusion for the treatment of degenerative lumbar disease in adults: a radiographic study. J Neurosurg Spine 2011;15:92-6. [Crossref] [PubMed]
- Ahmadian A, Bach K, Bolinger B, et al. Stand-alone minimally invasive lateral lumbar interbody fusion: multicenter clinical outcomes. J Clin Neurosci 2015;22:740-6. [Crossref] [PubMed]
- Anand N, Baron EM, Khandehroo B, et al. Long-term 2- to 5-year clinical and functional outcomes of minimally invasive surgery for adult scoliosis. Spine (Phila Pa 1976) 2013;38:1566-75. [Crossref] [PubMed]
- Anand N, Baron EM, Thaiyananthan G, et al. Minimally invasive multilevel percutaneous correction and fusion for adult lumbar degenerative scoliosis: a technique and feasibility study. J Spinal Disord Tech 2008;21:459-67. [Crossref] [PubMed]
- Benglis DM, Elhammady MS, Levi AD, et al. Minimally invasive anterolateral approaches for the treatment of back pain and adult degenerative deformity. Neurosurgery 2008;63:191-6. [Crossref] [PubMed]
- Caputo AM, Michael KW, Chapman TM, et al. Extreme lateral interbody fusion for the treatment of adult degenerative scoliosis. J Clin Neurosci 2013;20:1558-63. [Crossref] [PubMed]
- Caputo AM, Michael KW, Chapman TM Jr, et al. Clinical outcomes of extreme lateral interbody fusion in the treatment of adult degenerative scoliosis. Sci World J 2012;2012:680643.
- Castro C, Oliveira L, Amaral R, et al. Is the lateral transpsoas approach feasible for the treatment of adult degenerative scoliosis? Clin Orthop Relat Res 2014;472:1776-83. [Crossref] [PubMed]
- Dakwar E, Cardona RF, Smith DA, et al. Early outcomes and safety of the minimally invasive, lateral retroperitoneal transpsoas approach for adult degenerative scoliosis. Neurosurg Focus 2010;28:E8. [Crossref] [PubMed]
- Deukmedjian AR, Ahmadian A, Bach K, et al. Minimally invasive lateral approach for adult degenerative scoliosis: lessons learned. Neurosurg Focus 2013;35:E4. [Crossref] [PubMed]
- Deukmedjian AR, Dakwar E, Ahmadian A, et al. Early outcomes of minimally invasive anterior longitudinal ligament release for correction of sagittal imbalance in patients with adult spinal deformity. Sci World J 2012;2012:789698.
- Flouzat-Lachaniette CH, Ratte L, Poignard A, et al. Minimally invasive anterior lumbar interbody fusion for adult degenerative scoliosis with 1 or 2 dislocated levels. J Neurosurg Spine 2015;23:739-46. [Crossref] [PubMed]
- Haque RM, Uddin OM, Ahmed Y, et al. "Push-Through" Rod Passage Technique for the Improvement of Lumbar Lordosis and Sagittal Balance in Minimally Invasive Adult Degenerative Scoliosis Surgery. Clin Spine Surg 2016. [Epub ahead of print].
- Isaacs RE, Podichetty VK, Santiago P, et al. Minimally invasive microendoscopy-assisted transforaminal lumbar interbody fusion with instrumentation. J Neurosurg Spine 2005;3:98-105. [Crossref] [PubMed]
- Johnson RD, Valore A, Villaminar A, et al. Pelvic parameters of sagittal balance in extreme lateral interbody fusion for degenerative lumbar disc disease. J Clin Neurosci 2013;20:576-81. [Crossref] [PubMed]
- Karikari IO, Nimjee SM, Hardin CA, et al. Extreme lateral interbody fusion approach for isolated thoracic and thoracolumbar spine diseases: initial clinical experience and early outcomes. J Spinal Disord Tech 2011;24:368-75. [Crossref] [PubMed]
- Kelleher MO, Timlin M, Persaud O, et al. Success and failure of minimally invasive decompression for focal lumbar spinal stenosis in patients with and without deformity. Spine (Phila Pa 1976) 2010;35:E981-7. [Crossref] [PubMed]
- Khajavi K, Shen AY. Two-year radiographic and clinical outcomes of a minimally invasive, lateral, transpsoas approach for anterior lumbar interbody fusion in the treatment of adult degenerative scoliosis. Eur Spine J 2014;23:1215-23. [Crossref] [PubMed]
- Liu W, Chen XS, Jia LS, et al. The clinical features and surgical treatment of degenerative lumbar scoliosis: a review of 112 patients. Orthop Surg 2009;1:176-83. [Crossref] [PubMed]
- Manwaring JC, Bach K, Ahmadian AA, et al. Management of sagittal balance in adult spinal deformity with minimally invasive anterolateral lumbar interbody fusion: a preliminary radiographic study. J Neurosurg Spine 2014;20:515-22. [Crossref] [PubMed]
- Marchi L, Oliveira L, Amaral R, et al. Anterior elongation as a minimally invasive alternative for sagittal imbalance-a case series. HSS J 2012;8:122-7. [Crossref] [PubMed]
- Matsumura A, Namikawa T, Terai H, et al. The influence of approach side on facet preservation in microscopic bilateral decompression via a unilateral approach for degenerative lumbar scoliosis. Clinical article. J Neurosurg Spine 2010;13:758-65. [Crossref] [PubMed]
- Phillips FM, Isaacs RE, Rodgers WB, et al. Adult degenerative scoliosis treated with XLIF: clinical and radiographical results of a prospective multicenter study with 24-month follow-up. Spine (Phila Pa 1976) 2013;38:1853-61. [Crossref] [PubMed]
- Sclafani JA, Raiszadeh K, Raiszadeh R, et al. Validation and analysis of a multi-site MIS Prospective Registry through sub-analysis of an MIS TLIF Subgroup. Int J Spine Surg 2014;8.
- Tormenti MJ, Maserati MB, Bonfield CM, et al. Complications and radiographic correction in adult scoliosis following combined transpsoas extreme lateral interbody fusion and posterior pedicle screw instrumentation. Neurosurg Focus 2010;28:E7. [Crossref] [PubMed]
- Transfeldt EE, Topp R, Mehbod AA, et al. Surgical outcomes of decompression, decompression with limited fusion, and decompression with full curve fusion for degenerative scoliosis with radiculopathy. Spine (Phila Pa 1976) 2010;35:1872-5. [Crossref] [PubMed]
- Waddell B, Briski D, Qadir R, et al. Lateral lumbar interbody fusion for the correction of spondylolisthesis and adult degenerative scoliosis in high-risk patients: early radiographic results and complications. Ochsner J 2014;14:23-31. [PubMed]
- Wang MY. Improvement of sagittal balance and lumbar lordosis following less invasive adult spinal deformity surgery with expandable cages and percutaneous instrumentation. J Neurosurg Spine 2013;18:4-12. [Crossref] [PubMed]
- Wang MY, Mummaneni PV. Minimally invasive surgery for thoracolumbar spinal deformity: initial clinical experience with clinical and radiographic outcomes. Neurosurg Focus 2010;28:E9. [Crossref] [PubMed]
- Herkowitz HN, Kurz LT. Degenerative lumbar spondylolisthesis with spinal stenosis. A prospective study comparing decompression with decompression and intertransverse process arthrodesis. J Bone Joint Surg Am 1991;73:802-8. [PubMed]
- Aebi M. The adult scoliosis. Eur Spine J 2005;14:925-48. [Crossref] [PubMed]
- Grubb SA, Lipscomb HJ. Diagnostic findings in painful adult scoliosis. Spine (Phila Pa 1976) 1992;17:518-27. [Crossref] [PubMed]
- Smith JS, Shaffrey CI, Berven S, et al. Improvement of back pain with operative and nonoperative treatment in adults with scoliosis. Neurosurgery 2009;65:86-93; discussion 93-4. [Crossref] [PubMed]
- Glassman SD, Hamill CL, Bridwell KH, et al. The impact of perioperative complications on clinical outcome in adult deformity surgery. Spine (Phila Pa 1976) 2007;32:2764-70. [Crossref] [PubMed]
- Sansur CA, Smith JS, Coe JD, et al. Scoliosis research society morbidity and mortality of adult scoliosis surgery. Spine (Phila Pa 1976) 2011;36:E593-7. [Crossref] [PubMed]
- Schwab FJ, Lafage V, Farcy JP, et al. Predicting outcome and complications in the surgical treatment of adult scoliosis. Spine (Phila Pa 1976) 2008;33:2243-7. [Crossref] [PubMed]
- Smith JS, Sansur CA, Donaldson WF 3rd, et al. Short-term morbidity and mortality associated with correction of thoracolumbar fixed sagittal plane deformity: a report from the Scoliosis Research Society Morbidity and Mortality Committee. Spine (Phila Pa 1976) 2011;36:958-64. [Crossref] [PubMed]
- Isaacs RE, Hyde J, Goodrich JA, et al. A prospective, nonrandomized, multicenter evaluation of extreme lateral interbody fusion for the treatment of adult degenerative scoliosis: perioperative outcomes and complications. Spine (Phila Pa 1976) 2010;35:S322-30. [Crossref] [PubMed]
- Swank S, Lonstein JE, Moe JH, et al. Surgical treatment of adult scoliosis. A review of two hundred and twenty-two cases. J Bone Joint Surg Am 1981;63:268-87. [PubMed]
- Smith JS, Shaffrey CI, Berven S, et al. Operative versus nonoperative treatment of leg pain in adults with scoliosis: a retrospective review of a prospective multicenter database with two-year follow-up. Spine (Phila Pa 1976) 2009;34:1693-8. [Crossref] [PubMed]
- Le TV, Vivas AC, Dakwar E, et al. The effect of the retroperitoneal transpsoas minimally invasive lateral interbody fusion on segmental and regional lumbar lordosis. Scientific World Journal 2012;2012:516706. [PubMed]
- Pimenta L, Fortti F, Oliveira L, et al. Anterior column realignment following lateral interbody fusion for sagittal deformity correction. Eur J Orthop Surg Traumatol 2015;25 Suppl 1:S29-33. [Crossref] [PubMed]
- Saigal R, Mundis GM Jr, Eastlack R, et al. Anterior Column Realignment (ACR) in Adult Sagittal Deformity Correction: Technique and Review of the Literature. Spine (Phila Pa 1976) 2016;41 Suppl 8:S66-73. [PubMed]
- Baker JK, Reardon PR, Reardon MJ, et al. Vascular injury in anterior lumbar surgery. Spine (Phila Pa 1976) 1993;18:2227-30. [Crossref] [PubMed]
- Regan JJ, McAfee PC, Guyer RD, et al. Laparoscopic fusion of the lumbar spine in a multicenter series of the first 34 consecutive patients. Surg Laparosc Endosc 1996;6:459-68. [Crossref] [PubMed]
- Liu JC, Ondra SL, Angelos P, et al. Is laparoscopic anterior lumbar interbody fusion a useful minimally invasive procedure? Neurosurgery 2002;51:S155-8. [Crossref] [PubMed]
- Ozgur BM, Aryan HE, Pimenta L, et al. Extreme Lateral Interbody Fusion (XLIF): a novel surgical technique for anterior lumbar interbody fusion. Spine J 2006;6:435-43. [Crossref] [PubMed]
- Phan K, Rao PJ, Scherman DB, et al. Lateral lumbar interbody fusion for sagittal balance correction and spinal deformity. J Clin Neurosci 2015;22:1714-21. [Crossref] [PubMed]
Contributions: (I) Conception and design: K Phan, RJ Mobbs, F Altaf; (II) Administrative support: K Phan, RJ Mobbs, P McKenna, T Rajagopal, F Altaf; (III) Provision of study materials or patients: K Phan, RJ Mobbs, P McKenna, T Rajagopal, F Altaf; (IV) Collection and assembly of data: K Phan, YR Huo, JA Hogan, J Xu, A Dunn, SK Cho, F Altaf; (V) Data analysis and interpretation: All authors; (VI) Manuscript writing: All authors; (VII) Final approval of manuscript: All authors.


