
Minimally invasive spine surgery as treatment for persistent infectious lumbar spondylodiscitis: a systematic review and meta-analysis
Introduction
Infectious spondylodiscitis (IS) is a term used to encompass a variety of diseases including, but not limited to, spondylitis, discitis, spondylodiscitis, and vertebral osteomyelitis (1). Risk factors for IS include intravenous drug use, other serious medical comorbidities, advanced age, and an immunocompromised status (2,3). The incidence of IS has increased in recent years, which can largely be attributed to advancements in medical care that prolongs life expectancies for elderly and immunocompromised populations and increased rates of hospital-associated infections (2-4). Spinal surgeries are also being performed at an increasing rate, which further contributes to increasing rates of IS (4).
Conservative treatment is the standard of care for IS, and includes identification of the causative organism followed by long-term sensitive antibiotic administration and external immobilization (2-6). However, conservative management of IS has failure rates between 12% and 18% (7). Surgical interventions are typically reserved for cases of antibiotic resistant IS. In order to prevent the need for surgical intervention and progression of disease, it is extremely important to properly identify and treat the causative organism as early as possible (2-6).
Recent advances in minimally invasive surgery (MIS) have provided new alternatives to open surgery methods for treating resistant cases of IS (7). A previous study determined that MIS allows for sufficient debridement, alleviation of pain, and has a high pathogen identification rate (1). Because MIS is a relatively new treatment for resistant IS, outcome results are limited and the true benefit in comparison to open surgery is largely unknown. The purpose of this systematic review was to compare demographic and outcome data for MIS versus open surgery among patients with lumbar IS. We hypothesized that there will not be a significant difference in post-operative erythrocyte sedimentation rate (ESR), C-reactive protein (CRP), and visual analog scale (VAS) values between MIS and open surgery groups. We present the following article in accordance with the PRISMA reporting checklist (available at https://jss.amegroups.com/article/view/10.21037/jss-21-50/rc).
Methods
Literature search
In an attempt to identify all studies on MIS treatment for lumbar IS, a comprehensive literature search was performed in accordance with PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines. PubMed, Embase, and Scopus (last accessed 12/20/20) were utilized to search for the following terms: (“surgical management” OR “minimally invasive surgery”) AND (“lumbar spine infection” OR “infectious spondylodiscitis” OR “pyogenic spondylodiscitis” OR “vertebral osteomyelitis”). The search was narrowed to include only articles published between January 2015 through June 2021.
Inclusion and exclusion criteria
The inclusion criteria were articles published in English that reported ESR, CRP, and/or pain VAS scores before and after MIS or Open surgery. Exclusion criteria were non-English language articles, nonhuman studies, case reports, review articles, studies with less than 5 patients in their sample size, technique articles, and articles that did not report patient outcomes. One author performed the initial search, and two authors independently screened the search results to identify articles that met the inclusion criteria. The references of the included studies were further reviewed to identify any other relevant papers that may have been missed by the initial search.
Data collection
Two authors independently extracted data from each study and recorded the results on a standardized spreadsheet. Any differences with in the data collected between the two authors was reviewed by a third author. The following data was extracted: first author, year of publication, sample size, study design, patient age (years), patient sex (male/female), length of follow-up (months), type of surgery (MIS/open), ESR, CRP and VAS scores (preoperatively and postoperatively). For ESR, CRP, and VAS scores the mean difference from preoperative to postoperative were used as the primary comparison between the MIS and open groups. The Downs and Black study quality assessment tool was used for grading the methodological quality and risk for bias for each study (8). High quality/low risk of bias on the Downs and Black tool is indicated by a maximum score of 9 for case series. Level of evidence was determined based on the Center for Evidence-based Medicine criteria (9).
Statistical analysis
The pooled estimate of means (for continuous data) or proportions (for categorical data) were compared between the minimally invasive and open surgery groups. Studies were excluded from analysis if they did not provide an estimate of variance for a particular mean value. The Cochran Q statistic (significance level was P<0.05) and I2 (significance level was I2>50%) were calculated to test for heterogeneity of the data. Tests of heterogeneity were significant and random-effects models were subsequently used. Forest plots with means or proportions and corresponding 95% confidence intervals are provided. Comprehensive Meta-Analysis Software (Version 3; Biostat) was used for all analyses.
Results
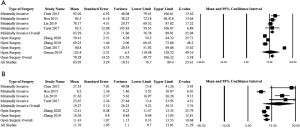
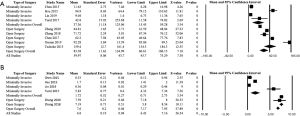
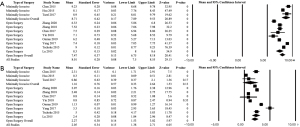
A total of 474 unique articles were identified upon initial search, of which 443 were excluded based on title and abstract. The remaining 31 articles were screened and of these, 17 were excluded and the remaining 14 were included in the final analysis (Figure 1). Thirteen of 14 studies that met the inclusion criteria were Level IV case series, and one was Level III (Table 1). There was no statistically significant difference in sex for minimally invasive surgery versus open surgery (43.0% females vs. 57.0% males, P=1.00). Mean ESR was greater for patients undergoing minimally invasive versus open surgery both preoperatively [83.29 (95% CI: 76.78, 89.81) vs. 78.28 (95% CI: 49.77, 106.79), P<0.0001] and postoperatively [19.27 (95% CI: 9.23, 29.31) vs. 11.43 (95% CI: 9.33, 13.53), P<0.0001] (Figure 2). Mean CRP was lower for patients undergoing minimally invasive surgery versus open surgery both preoperatively [37.37 (95% CI: 15.45, 59.28) vs. 83.38 (95% CI: 60.61, 106.15), P<0.0001] and postoperatively [1.72 (95% CI: 0.71, 2.73) vs. 7.56 (95% CI: 7.17, 7.95), P<0.0001] (Figure 3). Mean preoperative pain VAS was greater for patients undergoing minimally invasive versus open surgery [8.71 (95% CI: 7.89, 9.53) vs. 7.47 (95% CI: 6.75, 8.18), P<0.0001]; whereas mean postoperative pain VAS was lower for those undergoing minimally invasive surgery versus open surgery [1.16 (95% CI: −0.33, 2.64), vs. 2.27 (95% CI: 1.53, 3.02), P<0.0001] (Figure 4).

Table 1
| First author | Year of publication | Level of evidence | Study design | Downs and Black score | Type of surgery | Patients | |||
|---|---|---|---|---|---|---|---|---|---|
| Mean age years [range] | N | Male: female | Mean follow up, months | ||||||
| Chen HC (1) | 2015 | IV | Case series | 7 | MIS | 65.6 [49–84] | 13 | 5:8 | 3 |
| Hsu LC (2) | 2015 | IV | Case series | 7 | MIS | 57.8 [35–73] | 22 | 16:6 | 24 |
| Lu ML (3) | 2015 | IV | Case series | 6 | Open | 60.4 [37–86] | 28 | 13:15 | 18 |
| Tschöke SK (4) | 2015 | IV | Case series | 7 | Open | 74.3 [68–81] | 18 | 4:14 | 12 |
| Chen Y (5) | 2017 | IV | Case series | 8 | Open | 56.1 [46–66] | 24 | 11:13 | 24 |
| Turel MK (6) | 2017 | IV | Case series | 8 | MIS | 60.1 [55–69] | 7 | 5:2 | 4.5 |
| Yang SC (7) | 2017 | IV | Case series | 6 | Open | 53.5 [39–73] | 20 | 13:7 | 12 |
| Lin CY (10) | 2019 | IV | Case series | 8 | MIS | 60 [27–84] | 60 | 39:21 | 3 |
| Griffith-Jones W (11) | 2018 | III | Case series | 7 | MIS | 63 [51–82] | 10 | 7:3 | |
| Yin XY (12) | 2018 | IV | Case series | 6 | Open | 46.6 [35–56] | 16 | 12:4 | 35.3 |
| Zhang T (13) | 2018 | IV | Case series | 7 | Open | 45 [34–67] | 23 | 13:10 | 27 |
| Omran K (14) | 2019 | IV | Case series | 6 | Open | 46.6 [37–58] | 25 | 14:11 | 22 |
| Zhang HQ (15) | 2020 | IV | Case series | 6 | Open | 43.5 [32–56] | 27 | 16:11 | 35.7 |
| Zhou B (16) | 2020 | IV | Case series | 7 | Open | 55.7 [41–74] | 18 | 10:8 | 18 |
MIS, minimally invasive surgery.
Discussion
Our systematic review aimed to compare MIS with open surgery techniques for the treatment of antibiotic resistant IS. We found no significant difference in the ratio of male to female patients that were treated with MIS compared to open procedures. When comparing inflammatory markers, post-operative ESR was higher in the MIS group while post-operative CRP and VAS pain scores were greater in the open group. The findings do not align with our hypothesis as the two groups were not statistically similar in all measures. However, the MIS group showed favorable results for CRP and VAS values in comparison to the open group, which supports its use in this patient population. There have not been any previous studies comparing MIS to open surgery in patients with resistant IS, but there is evidence supporting MIS as an effective method for resistant spine infections in general. In addition, our results indicate that MIS provides comparable functional outcome to patients. Previous evidence has detailed that MIS techniques as a class offer patients shorter recovery time and lower complications rates when compared to an open approach (10). This combination offers viability for MIS being a reasonable alternative to open surgery for patients with resistant IS.
The incidence of IS has been on the rise in recent years due to several factors including increased hospital-associated infection rates and a higher number of spinal surgeries performed. Additionally, the rise in average life expectancy means a greater proportion of the population is immunosuppressed or suffering from other comorbid conditions. The combination of these population factors is associated with an increased incidence of IS as well as increased antibiotic resistance infections and a poorer disease course (2-7). Cases of IS that are resistant to conventional antibiotic and immobilization treatments are most concerning, especially in these high-risk populations (3,4). Despite the need, there is currently no standardized approach for treating cases of resistant IS. This poses a heightened risk of infection progression and deleterious effects, which can include neurological deficits and physical deformities as some of the possible outcomes (3,4). In the past, open surgical procedures have typically been used for treating resistant IS; however, recent data including this study support MIS techniques as a viable alternative that should be considered and further studied.
While our findings support the use of MIS as a comparable alternative to open surgery for patients with resistant IS, there needs to be further specification on which patients would be best suited for MIS treatment. Any patient where the infection has caused gross deformity or instability of the spinal column, i.e., empyema, would not be a candidate for an MIS approach and these patients would still require open surgery to regain stability through a fusion procedure. One study by Chen et al. 2017 estimated that between 19 and 25% of IS patients that required surgery would need anterior fusion (5,11). For patients without deformity or instability, we support the use of MIS approach. The evidence of statistically similar outcomes between MIS and open techniques along with the findings of Zarghooni et al. 2012, which found a significantly greater risk of post-operative infection with implantation, drives a compelling case in support of MIS in these patients (17).
The use of CT-guided biopsy was not a direct focus of this study, but it must be included in this discussion for a comprehensive review of all possible options. CT-guided biopsies are powerful tools that typically allow for easier sample access with less invasive procedures. In theory, this sounds like an ideal approach to obtain samples and target antibiotic therapy from infectious sites when compared with MIS, however, the rates of pathogen identification have been shown to be underwhelming with this approach. Previous evidence has found the culture rate with CT biopsy to less than 50% (6,12). In broad comparison we found the culture rate to be 76.1% in the MIS group. Moreover, CT biopsy does not have the benefit of removing adequate tissue, which can provide further therapeutic benefits (8). While there may be a role for CT biopsy in improving antibiotic management, it is not an appropriate option for patients that are resistant to antibiotics and require surgical intervention.
Importantly, it must be emphasized that surgery is not a first-line therapy for IS. It is typically reserved for cases that have proven refractory to first-line antibiotic therapy, which remains the foundation of IS management; 98% of IS cases have bacterial causes and 87% of all cases can be treated successfully with antibiotics alone (18). Guidelines from the Infectious Disease Society of American recommends broad spectrum antibiotics until the pathogen can be identified and then targeted therapy should be administered for 6 weeks. Serial ESR and CRP measurements should be used to monitor the response to antibiotics. This recommendation set the precedence for this study’s focus on monitoring surgical treatment through ESR and CRP. Reasons to escalate treatment from medical management to surgical considerations include, but are not limited to neurological deficits, spinal deformity or instability, and persistent infection after the duration of the recommended course.
This is the first systematic review, to our knowledge, that has compared the outcomes following MIS versus open surgery among patients with persistent IS. This systematic review was limited by the number of studies that reported clinical results of resistant IS, which resulted in a smaller sample size for some of the analyses. There was a significant difference in pre-operative ESR and CRP scores that could create bias in the results. Due to the size of the study we could not select studies that only had similar pre-operative inflammatory markers. All of the included studies except for 1 were Level IV case series (1 was Level III), but all studies scored high on the Downs and Black tool suggesting good quality and low risk for bias. We found no difference in sex between studies of MIS versus open surgery, however we were unable to compare other demographics (e.g., age) because the studies did not report an estimate of variance which is required for pooling data. We limited this analysis to peer-reviewed articles published in English, which may run the risk of publication and language bias.
Acknowledgments
Funding: None.
Footnote
Reporting Checklist: The authors have completed the PRISMA reporting checklist. Available at https://jss.amegroups.com/article/view/10.21037/jss-21-50/rc
Peer Review File: Available at https://jss.amegroups.com/article/view/10.21037/jss-21-50/prf
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://jss.amegroups.com/article/view/10.21037/jss-21-50/coif). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Chen HC, Huang TL, Chen YJ, et al. A Minimally Invasive Endoscopic Surgery for Infectious Spondylodiscitis of the Thoracic and Upper Lumbar Spine in Immunocompromised Patients. Biomed Res Int 2015;2015:780451. [Crossref] [PubMed]
- Hsu LC, Tseng TM, Yang SC, et al. Bilateral Portal Percutaneous Endoscopic Debridement and Lavage for Lumbar Pyogenic Spondylitis. Orthopedics 2015;38:e856-63. [Crossref] [PubMed]
- Lu ML, Niu CC, Tsai TT, et al. Transforaminal lumbar interbody debridement and fusion for the treatment of infective spondylodiscitis in the lumbar spine. Eur Spine J 2015;24:555-60. [Crossref] [PubMed]
- Tschöke SK, Fuchs H, Schmidt O, et al. Single-stage debridement and spinal fusion using PEEK cages through a posterior approach for eradication of lumbar pyogenic spondylodiscitis: a safe treatment strategy for a detrimental condition. Patient Saf Surg 2015;9:35. [Crossref] [PubMed]
- Chen Y, Yang JS, Li T, et al. One-stage Surgical Management for Lumbar Brucella Spondylitis by Posterior Debridement, Autogenous Bone Graft and Instrumentation: A Case Series of 24 Patients. Spine (Phila Pa 1976) 2017;42:E1112-8. [Crossref] [PubMed]
- Turel MK, Kerolus M, Deutsch H. The role of minimally invasive spine surgery in the management of pyogenic spinal discitis. J Craniovertebr Junction Spine 2017;8:39-43. [Crossref] [PubMed]
- Yang SC, Chen HS, Kao YH, et al. Single-stage anterior debridement and reconstruction with tantalum mesh cage for complicated infectious spondylitis. World J Orthop 2017;8:710-8. [Crossref] [PubMed]
- Downs SH, Black N. The feasibility of creating a checklist for the assessment of the methodological quality both of randomised and non-randomised studies of health care interventions. J Epidemiol Community Health 1998;52:377-84. [Crossref] [PubMed]
- Howick J, Chalmers I, Glasziou P, et al. The 2011 Oxford CEBM Levels of Evidence (Introductory Document). Available online: https://www.cebm.ox.ac.uk/resources/levels-of-evidence/levels-of-evidence-introductory-document
- Lin CY, Chang CC, Chen YJ, et al. New Strategy for Minimally Invasive Endoscopic Surgery to Treat Infectious Spondylodiscitis in the Thoracolumbar Spine. Pain Physician 2019;22:281-93. [PubMed]
- Griffith-Jones W, Nasto LA, Pola E, et al. Percutaneous suction and irrigation for the treatment of recalcitrant pyogenic spondylodiscitis. J Orthop Traumatol 2018;19:10. [Crossref] [PubMed]
- Yin XH, Liu ZK, He BR, et al. One-stage surgical management for lumber brucella spondylitis with anterior debridement, autogenous graft, and instrumentation. Medicine (Baltimore) 2018;97:e11704. [Crossref] [PubMed]
- Zhang T, Hu J, Wu J, et al. One-stage posterior debridement and fusion combined with irrigation and drainage for the treatment of postoperative lumbar spondylodiscitis. Acta Orthop Traumatol Turc 2018;52:277-82. [Crossref] [PubMed]
- Omran K, Ibrahim AH. Outcome of Transforminal Lumbar Thorough Debridement, Decompression, and Spondylodesis Technique in Treatment of 25 Patients with Pyogenic Spondylodiscitis. World Neurosurg 2019; [Epub ahead of print]. [Crossref] [PubMed]
- Zhang HQ, Wang YX, Wu JH, et al. Debridement and Interbody Graft Using Titanium Mesh Cage, Posterior Monosegmental Instrumentation, and Fusion in the Surgical Treatment of Monosegmental Lumbar or Lumbosacral Pyogenic Vertebral Osteomyelitis via a Posterior-Only Approach. World Neurosurg 2020;135:e116-25. [Crossref] [PubMed]
- Zhou B, Kang YJ, Chen WH. Continuous Epidural Irrigation and Drainage Combined with Posterior Debridement and Posterior Lumbar Inter-Body Fusion for the Management of Single-Segment Lumbar Pyogenic Spondylodiscitis. Surg Infect (Larchmt) 2020;21:262-7. [Crossref] [PubMed]
- Zarghooni K, Röllinghoff M, Sobottke R, et al. Treatment of spondylodiscitis. Int Orthop 2012;36:405-11. [Crossref] [PubMed]
- Berbari EF, Kanj SS, Kowalski TJ, et al. 2015 Infectious Diseases Society of America (IDSA) Clinical Practice Guidelines for the Diagnosis and Treatment of Native Vertebral Osteomyelitis in Adults. Clin Infect Dis 2015;61:e26-46. [Crossref] [PubMed]


