
Spinal subdural hematoma after interlaminar full-endoscopic decompression of lumbar spinal stenosis: a case report and literature review
Introduction
As a result of the development of minimally invasive surgery, endoscopic surgery for lumbar spinal stenosis has been widely used. There are many recent research studies about the effectiveness and safety of endoscopic decompression of spinal stenosis (1-3). Although most of these endoscopic procedures are performed safely, one of the rare complications is hematoma. In particular, spinal subdural hematoma (SSH) is a very rare complication, and there are only a few reports of SSH cases occurring after conventional open lumbar surgery (4-9). To our knowledge, there have been no case reports of SSH after endoscopic decompression. Herein, we describe a case of SSH that occurred after full-endoscopic interlaminar decompression and review the literature accordingly.
We present the following case in accordance with the CARE reporting checklist (available at http://dx.doi.org/10.21037/jss-20-664).
Case presentation
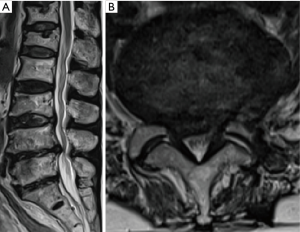
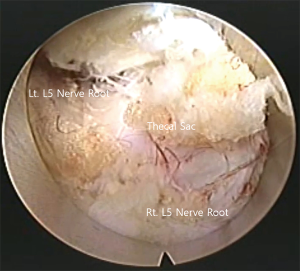
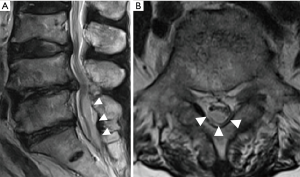
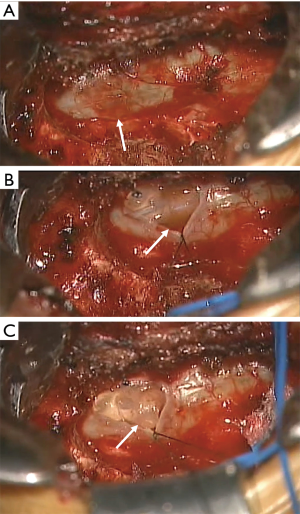
The patient was a 68-year-old woman with a 3-year history of radiating pain in her lower extremities. The neurological examination showed no motor grade impairment. She did not take any anticoagulant medications or have any underlying diseases. Her radiographs showed central canal and bilateral lateral recess stenosis with disc protrusion at the L4–5 level (Figure 1). Interlaminar full-endoscopic decompression was performed (Figure 2). Postoperatively, her radiating leg pain was improved (Figure 3). However, she began to complain of radiating pain on the right side 2 days after surgery. Physical therapy and pain medications were not effective. Furthermore, she had problems with urinary retention on postoperative day 7. Follow-up magnetic resonance imaging (MRI) was performed, and subdural fluid collection with compression of the cauda equina was observed at the index level (Figure 4). The fluid collection was T2 hyperintense, suggestive of a subdural hygroma or hematoma. We performed emergency open microscopic exploration at the index level and we found discolored dura matter without injury after L5 partial hemilaminectomy and flavectomy (Figure 5). Midline durotomy was conducted, and xanthrochromatic fluid flowed out immediately. After removing all of the fluid, we confirmed that the underlying arachnoid membrane was intact and pulsatile with no suspicious lesions. Then, the durotomy site was closed in a watertight fashion without cerebral spinal fluid leakage. The patient’s symptoms gradually resolved after exploration, and she was discharged without further complications (Figure 6).


All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Helsinki Declaration (as revised in 2013). Written informed consent was obtained from the patient.
Discussion
There are several causes of SSH, and they may be primary (spontaneous) or secondary to iatrogenic injury. In iatrogenic injuries, lumbar puncture, epidural anesthesia, and direct injury during surgery have been reported (10-12). The present patient developed an SSH postoperatively, whereas existing reports have described patients with a known dural injury (7,9) or those who have undergone open microscopic surgery (4-7,9) (Table 1). Our case is unique because it occurred after full-endoscopic decompression. In particular, decompression was performed without dural injury during the operation, and the patient’s symptoms improved immediately after surgery and then worsened. Therefore, the mechanism is questionable because the dura was intact in the operative field.

Full table
The mechanism underlying this complication is unknown. There are only a few hypotheses. First, injury of the bridging vessels between the dura and arachnoid membrane could be the most likely explanation for SSH without dural injury (13). The bridging vessels between the outer and inner layers of the dura mater may be the source of the bleed, although rare (6). Because there was no evident dural injury in our patient, we assume that dural manipulation during the decompressive procedure may have caused the hematoma formation. However, more unusually, it occurred at the level below not the level at which the surgery was performed. Therefore, more in-depth consideration of the mechanism is needed.
Second, the subdural space exists as a potential space between the arachnoid and dura mater, which is connected though a layer of border cells with weak, easily sheared intracellular connections (14). The shearing of these layers with minor trauma disrupts these weak junctions and splits open the space we consider as the subdural space. The hemorrhage was caused by the rupture of small extra-arachnoidal vessels located on the inner face of the dura mater. Bleeding may have subsequently developed and created a hematoma between the dura and intact arachnoid (8,14). As a result, this hypothesis is somewhat consistent. In the field of microscopic surgery, we also identified a safe arachnoid membrane.
Third, an unexpected increase in pressure in the abdominal or thoracic cavities could have elevated the pressure in the spinal vessels, particularly the valveless radiculomedullary veins because they cross the subdural and subarachnoid spaces (6).
These three hypotheses are theories that can arise spontaneously or after any surgery. Because this case of SSH developed after endoscopic decompression, there may be a unique mechanism caused by endoscopic surgery. A possible example is the pressure of the irrigation fluid. Persistent barotrauma may have caused these three mechanisms. Although there were no intraoperative complications and the patient’s condition improved immediately postoperatively, we also suspect the possibility of unrecognized injury to the dura mater. For example, we placed an epidural drain on the dura to prevent epidural hematoma. The tip of the drain may have compressed the dorsal dura, or the negative pressure of the drain may have also caused pressure change under the dura. We searched for literature on epidural drains and SSH, as well as literature on the location of appropriate epidural drains. Unfortunately, however, no literature has been found directly related to these. Therefore, we could not identify the exact reason for the delayed SSH after endoscopic decompression.
Emergency surgical decompression with hematoma evacuation is undisputed in SSH patients with neurological deficits (4,5,7,9,15). In contrast, patients without neurological deficits can be treated by conservative management (6,9) (Table 1). Although there were no problems during the operation, immediate postoperative MRI is needed if the postoperative neurologic deficit is aggravated or progressing compared to before the operation. Our patient had complete resolution of neurologic deficits after immediate decompression surgery. We recommend that surgeons should be cautious of this rare complication, and care should be taken to prevent delay of examination and treatment in similar patients.
SSH is a very rare complication of full-endoscopic spine decompressive surgery. Immediate radiologic evaluation is necessary for patients with postoperative severe pain or neurologic deficit, and emergency open evacuation with durotomy is the treatment of choice for patients with SSH and neurologic deterioration.
Acknowledgments
Funding: None.
Footnote
Reporting Checklist: The authors have completed the CARE reporting checklist. Available at http://dx.doi.org/10.21037/jss-20-664
Peer Review File: Available at http://dx.doi.org/10.21037/jss-20-664
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/jss-20-664). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Helsinki Declaration (as revised in 2013). Written informed consent was obtained from the patient.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Ito F, Ito Z, Shibayama M, et al. Step-by-step sublaminar approach with a newly-designed spinal endoscope for unilateral-approach bilateral decompression in spinal stenosis. Neurospine 2019;16:41. [Crossref] [PubMed]
- Yeung A, Roberts A, Zhu L, et al. Treatment of soft tissue and bony spinal stenosis by a visualized endoscopic transforaminal technique under local anesthesia. Neurospine 2019;16:52. [Crossref] [PubMed]
- Lee CW, Yoon KJ, Kim SW. Percutaneous Endoscopic Decompression in Lumbar Canal and Lateral Recess Stenosis–The Surgical Learning Curve. Neurospine 2019;16:63. [Crossref] [PubMed]
- Boe CC, Freedman BA, Kumar R, et al. Spinal subdural hematoma: a rare case of spinal subdural hematoma complicating routine, minimally invasive lumbar discectomy and decompression and relevant literature review. J Spine Surg 2017;3:112. [Crossref] [PubMed]
- Chang KC, Samartzis D, Luk KD, et al. Acute spinal subdural hematoma complicating lumbar decompressive surgery. Evid Based Spine Care J 2012;3:57-62. [Crossref] [PubMed]
- Gakhar H, Bommireddy R, Klezl Z, et al. Spinal subdural hematoma as a complication of spinal surgery: can it happen without dural tear? Eur Spine J 2013;22:S346-9. [Crossref] [PubMed]
- Gehri R, Zanetti M, Boos N. Subacute subdural haematoma complicating lumbar microdiscectomy. J Bone Joint Surg Br 2000;82:1042-5. [Crossref] [PubMed]
- Izeki M, Nagai K, Ota M, et al. Analysis of detailed clinical characteristics of spinal subdural hematoma following lumbar decompression surgery. J Orthop Sci 2018;23:857-64. [Crossref] [PubMed]
- Lykissas MG, Aichmair A, Herzog RJ, et al. Spinal subdural hematoma following lumbar decompressive surgery: a report of two cases. Wien Klin Wochenschr 2015;127:71-4. [Crossref] [PubMed]
- Edelson RN, Chernik NL, Posner JB. Spinal subdural hematomas complicating lumbar puncture: Occurrence in thrombocytopenic patients. Arch Neurol 1974;31:134-7. [Crossref] [PubMed]
- Jonsson L, Einarsson P, Olsson G. Subdural haematoma and spinal anaesthesia: a case report and an incidence study. Anaesthesia 1983;38:144-6. [Crossref] [PubMed]
- Russell NA, Benoit BG. Spinal subdural hematoma a review. Surg Neurol 1983;20:133-7. [Crossref] [PubMed]
- Miller DR, Ray A, Hourihan MD. Spinal subdural haematoma: how relevant is the INR? Spinal Cord 2004;42:477-80. [Crossref] [PubMed]
- Haines DE, Harkey HL, Al-Mefty O. The “subdural” space: a new look at an outdated concept. Neurosurgery 1993;32:111-20. [Crossref] [PubMed]
- Kobayashi K, Imagama S, Ando K, et al. Acute non-traumatic idiopathic spinal subdural hematoma: radiographic findings and surgical results with a literature review. Eur Spine J 2017;26:2739-43. [Crossref] [PubMed]


