Advantages and disadvantages of posterolateral approach for percutaneous endoscopic lumbar discectomy
Introduction
Percutaneous endoscopic lumbar discectomy (PELD) is one of the most sophisticated operative procedures for the treatment of lumbar disc herniation (LDH) (1-3). However, PELD has an anatomical limitation for endoscope insertion, and there are 3 different operative approaches: interlaminar, transforaminal, and posterolateral. Among these, the posterolateral approach (PLA) is most suitable for foraminal and extraforaminal LDH. Furthermore, PLA is most safely performed through “Kambin’s triangular working zone” (4); therefore, PELD beginners should first try this approach.
Foraminal and extraforaminal LDH frequently occur simultaneously. Furthermore, extraforaminal LDH sometimes combines with intra-canal LDH, with subsequent development of 2 radiculopathies (5). This pathological state is difficult to treat with a one-stage operation or an operation without facetectomy. PLA for PELD has a potential advantage in these complicated cases (3,6). However, the complications and limitations of PLA have not been fully described. We therefore analyzed cases treated with PLA for PELD in our hospital and provide useful information regarding the indications and limitations.
Methods
Twenty-nine consecutive patients with foraminal and extraforaminal LDH underwent PLA for PELD using a 7-mm diameter spinal full-endoscopic system (Richard Wolf GmbH, Knittlingen, Germany) between January 2014 and April 2016. All patients had lateral radiculopathy resistant to medical treatment, epidural steroids, and/or nerve block. As PLA for PELD offers significant surgical benefit for unsuccessful cases treated using a conventional posterior approach, we did not exclude patients who previously underwent discectomy at the same vertebral level. Similarly, we did not exclude patients with combined osseous foraminal stenosis, which can be enlarged using the foraminoplasty technique (7,8).
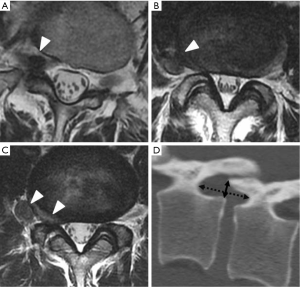
All patients underwent PLA for PELD at only 1 vertebral level. The LDH was classified into 3 types according to the location of the herniation on axial magnetic resonance imaging (MRI): foraminal, extraforaminal, and combined (Figure 1A,B,C). The foraminal height and width were calculated on sagittal views of preoperative computed tomography (CT) (Figure 1D). The height was measured as the maximum distance between the inferior margin of the pedicle of the superior vertebra and the superior margin of the pedicle of the inferior vertebra. The width was measured as the shortest distance between the superior edge of the superior articular process of the caudal vertebra and the posterior edge of inferior endplate of the cranial vertebra (9).
Patients were followed up for an average of 12.0 months (1–31 months) postoperatively. Pre- and postoperative leg pain was evaluated using the Numerical Rating Scale (NRS) score. Statistical analysis was performed using Student’s t-test. P values less than 0.05 were considered statistically significant. This study was approved by ethics committee of the Iwai Medical Foundation, and informed consent was obtained from the patients for publication of this study and any accompanying images.
Surgical technique
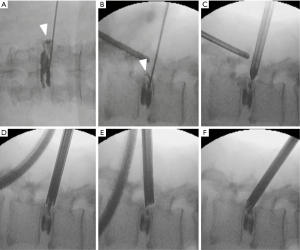
The patients were carefully log-rolled into the prone position. To enlarge the foramen, a pillow was placed between the operating table and anterior iliac crest. Except in an initial case (epidural anesthesia), the operations were performed under general anesthesia combined with motor evoked potential (MEP) monitoring to avoid intraoperative discomfort and postoperative piriformis syndrome (10). During the operation, a fluoroscope was placed across the center of the operating table to ensure appropriate positioning. An 8-mm skin and fascia incision was made 5–9 cm lateral from the midline at the target disc level under fluoroscopic guidance, and then an 18-gauge spinal needle was inserted into the disc. Discography with indigo carmine and a contrast medium was performed to stain the herniated disc material (Figure 2A,B). Following insertion of an obturator (Figure 2C), a 7-mm diameter working sheath was inserted. A 45° or 30° angled working sheath was placed on the disc surface to examine both the foraminal and extraforaminal spaces. Then, an endoscope (diameter of working channel: 4.1 mm) was inserted and the annulus, lateral part of the yellow ligament, and superior articular process (SAP) were confirmed. Generally, the herniated nucleus is located on the ventral side of the exiting nerve; we therefore explored the sequestered nucleus using several types of forceps and dissectors (Figure 2D,E,F) (Figure S1). When we found an annular tear, we also removed the damaged nucleus located under the annulus. After complete removal, the decompressed exiting nerve root becomes visible and freely movable. Finally, the annular tear and evacuated cavities were electrocoagulated using a bipolar radio-frequency electrode system (Elliquence, Baldwin, NY, USA), followed by careful endoscopic examination for epidural bleeding and hemostasis. After decompression, the working sheath was carefully removed, and skin was closed with a single suture.

Results
Of 29 patients registered for this study, 17 underwent PLA without foraminoplasty and 12 underwent PLA with foraminoplasty. The mean age was 56.8 years (range: 25–83 years). There were 21 male and 8 female. The most affected vertebral levels were L3/4 (13 cases) and L4/5 (13 cases). Each location of LDH predicted by preoperative MRI is shown in Table 1. The mean foraminal height was 17.6 mm (range, 13–23 mm), and the mean foraminal width was 8.6 mm (range, 7–13 mm). Within these ranges of foraminal size, the operative procedure of PLA was not restricted. The mean operative time was 37.1 min (range, 22–105 mim), the mean postoperative hospital stay was 2.4 days (range, 1–16 days), and blood loss was negligible in all patients (Table 1).
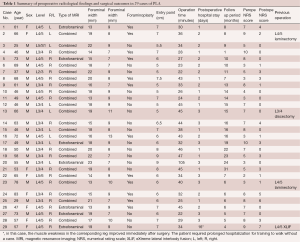
Full table
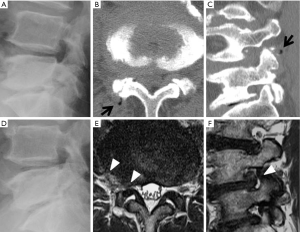
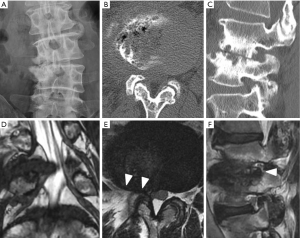
The mean NRS scores improved significantly from 6.1±2.4 to 1.8±2.0 (P<0.01). During the follow-up period, 1 patient (case 8) has not shown any improvement in NRS score. This patient required reoperation, and we performed microendoscopic discectomy (MED) 7 months after PELD. Although intraoperative findings did not show apparent recurrence or massive residual nucleus, we carefully removed scar tissue around the exiting nerve and enlarged the foramen. After the second operation, the patient only showed slight improvement in leg pain. Although preoperative plain roentgenography did not indicate any instability, MRI and CT showed a “facet fluid sign” (11,12) and “vacuum phenomenon” (13), respectively (Figure 3). We retrospectively speculated that the patient’s leg pain might be due to instability at the L4/5 disc level.
We also observed early recurrence in 1 patient (case 4) who presented with local scoliosis (coronal Cobb angle =13°) (Figure 4). He underwent a second operation 5 months after initial surgery. In this case, we assumed that the local scoliosis participated in the recurrence, and performed lumbar interbody fusion of the L3/4 vertebrae. In the 5 months after the second operation, he has not complained of any leg pain. We have not observed recurrences in other patients during the follow-up period in this study. We also did not observe intra- or postoperative complication in any patients. Furthermore, we performed PLA for PELD in 4 patients who previously underwent spinal surgery at the same vertebral level. The operative outcome was satisfactory, even in these cases.
Discussion
Foraminal or extraforaminal LDH extending into or beyond the foraminal zone accounts for 7–12% of all LDH cases (14,15). It has also been reported that the frequency of occurrence is nearly equal at the L4/5 and L3/4 levels (15). Furthermore, patients with foraminal or extraforaminal LDH are typically in their mid-fifties, ranging from 50–78 years of age (15). We also observed the same distributions in this study. Several different surgical techniques have been developed for foraminal or extraforaminal LDH (15,16), but PELD is the most recent and promising minimally invasive operative procedure.
Generally, facetectomy is not necessary for endoscopic removal of an extraforaminal LDH, although a small facetectomy (involving only a small part of the SAP, a so-called foraminoplasty) is occasionally a good surgical option for the endoscopic removal of foraminal or combined LDH. As foraminal or extraforaminal LDH predominantly occurs in elderly patients, and is complicated by lumbar degenerative disease such as facet joint osteoarthritis (15), removal of SAP osteophytes is sometimes required for entry of the endoscope. On the other hand, conventional open surgery occasionally requires a wider facetectomy, and the risk of postoperative instability increases (15). Anatomical cadaver studies clearly indicated that more medial access to Kambin’s triangle by foraminoplasty not only provides safe access to the intra-canal or foraminal space but also makes it possible to prevent exiting nerve injury (17). Therefore, foraminoplasty (removal of SAP from outside to medial, thereby enabling endoscopic access to the medial part of the triangle) using a surgical burr or trephine is an appropriate option in PLA for PELD. Furthermore, foraminoplasty does not increase the risk of instability.
The “hand-down technique” proposed by Sairyo et al. is another option (2). In this technique, downward pressure on the proximal side of the endoscope makes it possible to observe a more interior region through the foramen, thereby enabling removal of a nucleus that is extremely protruded toward the spinal canal. As continuous downward pressure on the endoscope might compress the exiting nerve, we should only use this technique for a short time, and repeat it after a long interval. In this study, we performed foraminoplasty in 12 cases, and used the hand-down technique in all cases to at least confirm the decompression of the foraminal space. The combination of PLA with foraminoplasty or hand-down technique seems to be appropriate for the treatment of foraminal or combined LDH.
PELD offers the advantages of small incision size (8 mm), rapid recovery, short hospital stay, limited blood loss, less destruction of surrounding tissues, and less postoperative pain (18). However, contraindications and inadequate indications have not been fully reported (19-21). In this study we showed that a narrow foramen (foraminal height ≥13 mm, foraminal width ≥7 mm) is not a limiting factor in PLA for PELD; however, we observed early recurrence in 1 patient who presented with local scoliosis (coronal Cobb angle =13°), and in 1 patient with suspected instability who showed no improvement in the NRS score. We subsequently excluded patients with local scoliosis (coronal Cobb angle >10°) and signs of instability such as a facet fluid sign (11,12) and vacuum phenomenon (13) on MRI and CT, respectively.
It is important to emphasize the importance of training and surgical experience, as PELD surgery does not have a steep learning curve, in contrast to MED (22,23). Early in training, operative complications such as exiting nerve injury can easily occur (24,25). Working sleeve insertion is harder to perform in the intra-canal region than in the extra-canal region. Furthermore, operative manipulation of the endoscope and forceps require delicate surgical skills because of the narrow operative field (4.1 mm diameter) and two-dimensional visualization. PLA for extraforaminal LDH is a simpler and safer procedure than other PELD approaches from the viewpoint of anatomy and technique. Therefore, we recommend performing PLA for PELD in the early period of training. We also recommend a short, intensive course of training on the use of the free-hand endoscope system under expert supervision to prevent intra-operative complications of PELD.
Conclusions
Preliminary results during a short follow-up period show that PLA for PELD is feasible for the treatment of foraminal and/or extraforaminal LDH with foraminal height ≥13 mm and foraminal width ≥7 mm. The procedure is effective for preservation of the facet joint; however, the indications must be carefully considered when local scoliosis and/or instability are present.
Acknowledgements
We would like to thank all the operating room staff for their technical assistance, and the medical records clerks who helped collect patient data. We would also like to thank all radiological department staff for accumulating CT and MRI data. This work was partly supported by a grant from the Iwai Medical Foundation.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: This study was approved by ethics committee of the Iwai Medical Foundation, and informed consent was obtained from the patients for publication of this study and any accompanying images.
References
- Choi G, Lee SH, Deshpande K, et al. Working channel endoscope in lumbar spine surgery. J Neurosurg Sci 2014;58:77-85. [PubMed]
- Sairyo K, Egawa H, Matsuura T, et al. State of the art: Transforaminal approach for percutaneous endoscopic lumbar discectomy under local anesthesia. J Med Invest 2014;61:217-25. [Crossref] [PubMed]
- Yeung AT. The Evolution and Advancement of Endoscopic Foraminal Surgery: One Surgeon's Experience Incorporating Adjunctive Techologies. SAS J 2007;1:108-17. [Crossref] [PubMed]
- Kambin P, Sampson S. Posterolateral percutaneous suction-excision of herniated lumbar intervertebral discs. Report of interim results. Clin Orthop Relat Res 1986.37-43. [PubMed]
- Paolini S, Ciappetta P, Raco A, et al. Combined intra-extracanal approach to lumbosacral disc herniations with bi-radicular involvement. Technical considerations from a surgical series of 15 cases. Eur Spine J 2006;15:554-8. [Crossref] [PubMed]
- Choi KC, Park CK. Percutaneous Endoscopic Lumbar Discectomy for L5-S1 Disc Herniation: Consideration of the Relation between the Iliac Crest and L5-S1 Disc. Pain Physician 2016;19:E301-8. [PubMed]
- Choi G, Lee SH, Lokhande P, et al. Percutaneous endoscopic approach for highly migrated intracanal disc herniations by foraminoplastic technique using rigid working channel endoscope. Spine (Phila Pa 1976) 2008;33:E508-15. [Crossref] [PubMed]
- Lee CW, Yoon KJ, Ha SS, et al. Foraminoplastic Superior Vertebral Notch Approach with Reamers in Percutaneous Endoscopic Lumbar Discectomy: Technical Note and Clinical Outcome in Limited Indications of Percutaneous Endoscopic Lumbar Discectomy. J Korean Neurosurg Soc 2016;59:172-81. [Crossref] [PubMed]
- Korovessis PG, Stamatakis MV, Baikousis AG. Reciprocal angulation of vertebral bodies in the sagittal plane in an asymptomatic Greek population. Spine (Phila Pa 1976) 1998;23:700-4; discussion 704-5. [Crossref] [PubMed]
- Kim JE, Kim KH. Piriformis syndrome after percutaneous endoscopic lumbar discectomy via the posterolateral approach. Eur Spine J 2011;20:1663-8. [Crossref] [PubMed]
- Hipp JA, Guyer RD, Zigler JE, et al. Development of a novel radiographic measure of lumbar instability and validation using the facet fluid sign. Int J Spine Surg 2015;9:37. [PubMed]
- Caterini R, Mancini F, Bisicchia S, et al. The correlation between exaggerated fluid in lumbar facet joints and degenerative spondylolisthesis: prospective study of 52 patients. J Orthop Traumatol 2011;12:87-91. [Crossref] [PubMed]
- Gellhorn AC, Katz JN, Suri P. Osteoarthritis of the spine: the facet joints. Nat Rev Rheumatol 2013;9:216-24. [Crossref] [PubMed]
- Epstein NE. Evaluation of varied surgical approaches used in the management of 170 far-lateral lumbar disc herniations: indications and results. J Neurosurg 1995;83:648-56. [Crossref] [PubMed]
- Epstein NE. Foraminal and far lateral lumbar disc herniations: surgical alternatives and outcome measures. Spinal Cord 2002;40:491-500. [Crossref] [PubMed]
- Pirris SM, Dhall S, Mummaneni PV, et al. Minimally invasive approach to extraforaminal disc herniations at the lumbosacral junction using an operating microscope: case series and review of the literature. Neurosurg Focus 2008;25:E10. [Crossref] [PubMed]
- Xin G, Shi-Sheng H, Hai-Long Z. Morphometric analysis of the YESS and TESSYS techniques of percutaneous transforaminal endoscopic lumbar discectomy. Clin Anat 2013;26:728-34. [Crossref] [PubMed]
- Li X, Hu Z, Cui J, et al. Percutaneous endoscopic lumbar discectomy for recurrent lumbar disc herniation. Int J Surg 2016;27:8-16. [Crossref] [PubMed]
- Ahn Y, Lee HY, Lee SH, et al. Dural tears in percutaneous endoscopic lumbar discectomy. Eur Spine J 2011;20:58-64. [Crossref] [PubMed]
- Choi G, Kang HY, Modi HN, et al. Risk of developing seizure after percutaneous endoscopic lumbar discectomy. J Spinal Disord Tech 2011;24:83-92. [Crossref] [PubMed]
- Cho JY, Lee SH, Lee HY. Prevention of development of postoperative dysesthesia in transforaminal percutaneous endoscopic lumbar discectomy for intracanalicular lumbar disc herniation: floating retraction technique. Minim Invasive Neurosurg 2011;54:214-8. [Crossref] [PubMed]
- Lee DY, Lee SH. Learning curve for percutaneous endoscopic lumbar discectomy. Neurol Med Chir (Tokyo) 2008;48:383-8; discussion 388-9. [Crossref] [PubMed]
- Wang H, Huang B, Li C, et al. Learning curve for percutaneous endoscopic lumbar discectomy depending on the surgeon's training level of minimally invasive spine surgery. Clin Neurol Neurosurg 2013;115:1987-91. [Crossref] [PubMed]
- Wang H, Zhou Y, Li C, et al. Risk factors for failure of single-level percutaneous endoscopic lumbar discectomy. J Neurosurg Spine 2015;23:320-5. [Crossref] [PubMed]
- Ahn SS, Kim SH, Kim DW. Learning Curve of Percutaneous Endoscopic Lumbar Discectomy Based on the Period (Early vs. Late) and Technique (in-and-out vs. in-and-out-and-in): A Retrospective Comparative Study. J Korean Neurosurg Soc 2015;58:539-46. [Crossref] [PubMed]
- Yokosuka J, Oshima Y, Kaneko T, et al. Advantages and disadvantages of posterolateral approach for percutaneous endoscopic lumbar discectomy. Asvide 2016;3:389. Available online: http://www.asvide.com/articles/1158


