
Cost analysis comparison between conventional microsurgical decompression and full-endoscopic interlaminar decompression for lumbar spinal stenosis surgery
Introduction
Lumbar spinal stenosis (LSS) is one of the commonest spine conditions faced worldwide especially in the over 65 age group (1). According to the Framingham study, degeneration of the lumbar spine occurs in 20–25% of the general population and increases beyond the over 50 age group (2). This trend is on the rise with estimated 23–25% of the population being older than 65 years (3,4). LSS can originate from developmental or degenerative causes. Developmental spinal stenosis which is a result of maldevelopment of the spinal elements may lead to multiple level involvement (5-8). More commonly, symptoms arise as a direct consequence of degenerative processes such as disc herniation, facet joint and ligamentum flavum hypertrophy and osteophytes. The degenerative cascade begins with disc degeneration followed by facet joint overload (9,10). Ligamentum flavum changes occur as a result leading to spinal stenosis and nerve compression. In the late stage, spondylolisthesis occurs which may cause spinal instability and deformity (11-13).
Patients with LSS requiring operation generally have good clinical response after decompression surgery (14-16). However, there is still no clear better surgical approach for LSS. Many options are available including open laminotomy, laminectomy, endoscopic assisted procedures or interspinal spacers (17). Two of the most commonly used techniques include full-endoscopic interlaminar decompression and conventional microsurgical decompression have had comparative randomized controlled trials (18,19). However, both procedures appear to have similar clinical and radiological outcomes.
In this modern age, the concern for raised healthcare costs (20) influences the type of treatment that is available to us as healthcare providers. Health economic evaluation is a necessary component for decision making in spine surgery. Accumulative costs to patients and the healthcare infrastructure are real concerns for the successful implementation of a new treatment into routine practice. Support for these new endeavors requires cost analysis data so that we can provide the best care at low costs (21,22). As such, the cost data for different surgical procedures for LSS is lacking and this study aims to address this limitation by a cost analysis comparison between full-endoscopic interlaminar decompression (MIS) and conventional microsurgical decompression (CD).
We present the following article in accordance with the CHEERS reporting checklist (available at http://dx.doi.org/10.21037/jss-20-552).
Methods
This study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). Because of the retrospective nature of the study and the collection of unit cost of items for the cost simulation modelling did not involve individual patients, the ethics committee waived the need for informed consent and ethics approval.
Surgical procedures
The CD surgery was a bilateral laminotomy technique. After the skin incision and exposure of the fascia, the musculature was prepared with a Caspar retractor inserted. A microscope was used for assistance during the decompression by cranial and caudal laminotomy, medial facetectomy and flavum resection. Then the retractor is placed at the opposite side for contralateral decompression. For the MIS approach, we utilized a unilateral access via a paramedian skin incision. Blunt dissection with a dilator is used towards the interlaminar window. The operation sheath is placed over the dilator with the beveled opening directed medially toward the ligamentum flavum. The surgery is performed under constant irrigation. The ipsilateral decompression is performed by cranial and caudal laminotomy, medial facetectomy and ligamentum flavum resection. Contralateral decompression is performed dorsal to the dura. Bony decompression is done first for cranial and caudal laminotomy and medial facetectomy followed by removal of the ligamentum flavum as well. In both procedures, surgery is complete when the dura and spinal nerves are decompressed. No disc procedures were performed.
Model structure and setting
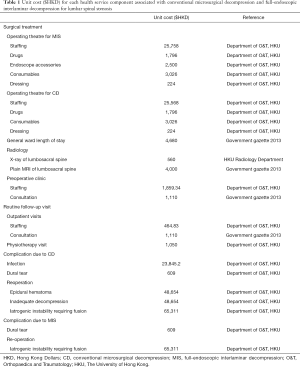
A decision analysis model comparing MIS to CD for patients with LSS over a one-year time horizon was conducted. This time horizon was selected as 1-year postoperative outcome was commonly evaluated in the trials or cohort studies comparing surgical approaches for patients for LSS. An executable model was built in Microsoft Excel. We simulated a cohort of average-risk patients attending for an index decompression procedure for LSS. All patients, regardless of undergoing MIS or CD were assessed for unit costs (Table 1) for operating theater, length of hospitalization, use of imaging, subsequent outpatient visits, and dealing with complications conditioning on the type of surgery for LSS. Since all operations of both CD and MIS surgery were of single-level, there was no additional fee associated with any multilevel decompression. All costs were listed in Hong Kong Dollars (HKD).
Full table
Relevant unit costs were retrieved from the Department of Orthopaedics and Traumatology, the University of Hong Kong at its affiliated hospitals: The Duchess of Kent Children’s Hospital and Queen Mary Hospital. Costs associated with radiology, hospitalizations, outpatient, and physiotherapy visits were based on the latest price list which itemized the charges to non-Hong Kong residents for use of health services in the hospital authority, as published in the 2013 government gazette (23).
Statistical analysis
Unit costs of the surgical procedure related to MIS and open surgery were estimated from actual hospital expenditure with respect to staffing (surgeons, anesthetists, nursing, and technical support staff), prescription of drugs (antibiotics and anesthesia), endoscope accessories, radiology (X-ray and plain MRI of lumbosacral spine), intraoperative consumables, and dressing categories.
Staff costs related to the operating theatre for surgery were calculated based on the number of hours spent immediate preoperatively, during surgery and postoperatively by each staff multiplied by respective hourly wages, as provided by the local hospital authority. After discharge from hospital, routine outpatient follow-up was included for both operations including one specialist outpatient visit for wound care 2 weeks after surgery, an additional visit for outcome assessment and complication screening, and one physiotherapy visit.
The risk of complication was calculated for the index operation with the risk of reoperation due to major complications namely epidural hematoma, inadequate decompression, and iatrogenic instability requiring fusion. Complications accounted for in this study included infection, dural tear, epidural hematoma, inadequate decompression, and iatrogenic instability requiring fusion. Of these, infection and dural tear were not considered for reoperation as they can be managed by other measures, but their respective cost for mediation were calculated. Estimated risks of individual surgical complications were sourced from two prospective randomized controlled trials (RCT) (18,19) studying MIS and CD treatment for LSS.
The average total costs associated with each MIS and CD surgery were compared based on the accumulative costs from surgery, complications and follow-up visits. The cost of complications was estimated by the unit cost of each complication and their respective complication rate as described in Table 2.

Full table
Results
There were 161 patients in the CD group and 160 patients in the MIS group. The mean age of patients was the same (63 years) for both groups. There were 83 females and 78 males for CD, and 82 females and 78 males for MIS. The mean operating time was 38 (range, 28–64) minutes for CD and 56 (range, 32–93) minutes for MIS. All operations were performed by 2 specialist surgeons with many years of experience. The surgeons were experienced in both methods and thus we do not consider the learning curve or surgeon inexperience as reasons for the complications in this study.
The unit costs of the surgical procedure related to MIS and open surgery were amounted to HKD$33,304 for MIS and HKD$30,614 for CD, estimated from the actual hospital expenditure. Through this simulation, the average total costs were HKD$54,863 for MIS and HKD$52,748 for CD (Table 3). With the general ward hospitalization, radiology and routine follow-up visits being of the same cost for both surgical approaches, the 3.9% (HKD$2,115) difference in total cost was largely due to differences in the unit cost of surgery and complications. For the unit cost of running one operating session, MIS costs 5.7% (HKD$2,690) more than CD. The accumulated surgical complication rates were 3.8% for MIS and 7.5% for CD. Hence, CD was 28.1% (HKD$575) costlier than MIS. Total number of hours spent by all staff per MIS and CD operation were 38.95 and 37.20, respectively. The breakdown of each MIS surgery was 5.0 hours for 2 surgeons, 10.45 hours for 2 anesthetists, 14.75 hours for 4 nurses, and 8.25 hours for 3 technical support staff. The breakdown for each CD surgery was 5.0 hours for 2 surgeons, 10.45 hours for 2 anesthetists, 14.5 hours for 4 nurses, and 7.25 hours for 3 technical support staff. Additional costs accrued for each operation (both MIS and CD) included 2 days of general ward stay, X-ray and MRI of the lumbosacral spine, and preoperative assessment by anesthetist, surgeon, and support staff.

Full table
Estimated risks of individual surgical complications are shown in Table 2. For infection, 2 weeks of antibiotic treatment (HKD$221.2), dressing (HKD$224) and 5 additional days of ward stay added up to HKD$23,845.2. Dural tears were managed intraoperatively with a dural patch (Tachosil® Fibrin Sealant patch) without the need for additional surgery. Hence, the only cost was for the dural patch (HKD$609). Epidural hematoma and inadequate decompression were only observed in CD. An additional surgery was required for clot evacuation and wider decompression. This cost (HKD$48,654) was estimated to be the same as another CD operation including an additional plain MRI for diagnosis and 4 additional days of ward stay. Iatrogenic instability requiring fusion was observed in both operations and the costs accrued were HKD$65,311 generated by an additional surgery (CD), use of intraoperative imaging, and for a single level lumbar fusion. For instrumented fusion, costs were referenced from Medtronic® CD Horizon® Legacy™ system. This included 4 pedicle screws (HKD$3,441 per screw), 1 rod (HKD$1,545) and 4 set screws (HKD$337 per item).
Discussion
In the current climate of increasing concerns regarding healthcare costs, health economics have an important role in surgical decision making on how information on costs, benefits and risks of MIS and CD procedures are incorporated prior to LSS surgery. Our study suggests that both MIS and CD procedures for LSS have important cost concerns. Despite both procedures having overall good clinical outcomes and low complication rates, costs from intraoperative consumables and revision fusion surgery results in a slight inferiority of MIS compared to CD.
The baseline cost comparison between the two procedures are similar due to an overall similar surgical procedure. However, there is an overall increase in surgical cost with MIS due to the consumables used for endoscopy. CD only requires an operating microscope with a single use sterile drape. The endoscopy conversely requires specific set of instruments which require routine cleansing and sterilization. Hence, each surgery increases the costs accrued for MIS. In fact, the fundamental differences in surgical approaches can introduce differences in related costs. Utilizing lumbar spinous process-splitting laminectomy instead of bilateral laminotomies may influence the overall cost-effectiveness of surgery. The unilateral approach with the lumbar spinous process-splitting laminectomy may reduce the overall surgical time and also the amount of muscle dissection and potential back pain postoperatively. This is important considering that the CD surgery is already shorter than MIS and the staff cost per hour is significant. However, the differences may not be too dramatic as the surgical difficulty is not increased and complication rates may not change much. Traditional full open laminectomy may be even quicker but is not a good comparison as most surgeons would consider using less invasive options nowadays.
Based on the complication rates described in the two trials (18,19), the effect of intraoperative costs is minimized as MIS has an overall lower complication rate than CD. This is reflected by a lower overall cost of complications in MIS. Yet, this difference is small and unable to overcome the cost of surgery. Although CD may have more cases requiring revision decompression due to epidural hematoma or inadequate decompression, iatrogenic instability is more prevalent for MIS. Since more cases require instrumented fusion in MIS, the incurred cost of implants balance out the overall costs of complications between the two approaches. As such, the cost of a reoperation due to epidural hematoma or inadequate decompression (HKD$48,654) pales in comparison to a fusion surgery (HKD$65,311). Although inadequate decompression is more related to the surgeon’s technique rather than an inherent problem of the surgical procedure, the overall rate of this complication is low. Increased complication of iatrogenic instability in MIS may be related to the visibility intraoperatively. In addition, the size of the spinal canal should also be considered. The reoperation rate has been reported to be 21.7% for patients with a narrowed canal (24). As CD is ultimately an open procedure, it can be argued that the anatomical structures are more clearly seen thus avoiding too much removal of the pars interarticularis or medial facet joint leading to iatrogenic instability.
The main limitation for this study is the use of complication data from the two randomized controlled trials (18,19). Both studies were performed by experienced endoscopic surgeons and thus the complication rates were low. This study has not taken into account of the learning curve effect of endoscopic surgery. As such, the low complication rates seen in the MIS group is likely an underestimation. Despite the overall costs are still in favor of CD as compared to MIS, the comparison is only modest. Further study comparing different levels of surgical experience may be useful to elucidate this effect. Also, this study did not examine the setting of an outpatient ambulatory surgery center. There can be differences in costs from admitting patients for surgeries as compared to those performed at ambulatory surgery centre, mainly in terms of costs incurred during hospitalization, whereas mobilizing microscopic instrument and its maintenance can also incur extra expenses. Regional variations should also be taken into consideration. For the costs incurred in our locality, a reference can be taken from early onset scoliosis surgery (25). Furthermore, the quality of life and health utility data were not available from published data from observational studies and clinical trials. Current evidence was uncertain to contribute to the estimation of quality-adjusted life-years, an essential outcome measure for cost-effectiveness and cost-utility analyses (26).
Conclusions
This is the first cost analysis study examining different techniques of LSS surgery and our findings have significant implications on decision making, and on how clinician and patient preference for surgical approach are determined on the basis of costs, benefits and risks. Debate of superiority between different operative procedures is frequent over the past decade. With newer and possibly more sophisticated techniques emerging in the spine market, it is important for us to gauge not only their clinical benefit but their associated healthcare costs. Our study suggests that there is a small cost saving benefit in short-term when performing CD as compared to MIS, and this estimation is likely an underestimation considering the learning curve effect of endoscopic surgery. With similar clinical outcomes observed from the referenced trials, there is no obvious health benefit of either procedure. Surgeons are justified to choose either procedure they are comfortable with as long as adequacy of decompression is achieved.
Acknowledgments
Funding: This study was financially supported by the Sanming Project of Medicine (SZSM201612055) “Team of Excellence in Spinal Deformities and Spinal Degeneration Diseases”.
Footnote
Reporting Checklist: The authors present the study in accordance with the CHEERS reporting checklist. Available at http://dx.doi.org/10.21037/jss-20-552
Data Sharing Statement: Available at http://dx.doi.org/10.21037/jss-20-552
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (Available at http://dx.doi.org/10.21037/jss-20-552). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). Because of the retrospective nature of the study and the collection of unit cost of items for the cost simulation modelling did not involve individual patients, the ethics committee waived the need for informed consent and ethics approval.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Deyo RA, Mirza SK, Martin BI, et al. Trends, major medical complications, and charges associated with surgery for lumbar spinal stenosis in older adults. JAMA 2010;303:1259-65. [Crossref] [PubMed]
- Kalichman L, Cole R, Kim DH, et al. Spinal stenosis prevalence and association with symptoms: the Framingham Study. Spine J 2009;9:545-50. [Crossref] [PubMed]
- Ammendolia C, Stuber K, de Bruin LK, et al. Nonoperative treatment of lumbar spinal stenosis with neurogenic claudication: a systematic review. Spine 2012;37:E609-16. [Crossref] [PubMed]
- Deyo RA. Treatment of lumbar spinal stenosis: a balancing act. Spine J 2010;10:625-7. [Crossref] [PubMed]
- Cheung JP, Samartzis D, Shigematsu H, et al. Defining clinically relevant values for developmental spinal stenosis: a large-scale magnetic resonance imaging study. Spine 2014;39:1067-76. [Crossref] [PubMed]
- Cheung JP, Shigematsu H, Cheung KM. Verification of measurements of lumbar spinal dimensions in T1- and T2-weighted magnetic resonance imaging sequences. Spine J 2014;14:1476-83. [Crossref] [PubMed]
- Cheung JPY, Ng KKM, Cheung PWH, et al. Radiographic indices for lumbar developmental spinal stenosis. Scoliosis Spinal Disord 2017;12:3. [Crossref] [PubMed]
- Cheung PWH, Tam V, Leung VYL, et al. The paradoxical relationship between ligamentum flavum hypertrophy and developmental lumbar spinal stenosis. Scoliosis Spinal Disord 2016;11:26. [Crossref] [PubMed]
- Buckwalter JA. Aging and degeneration of the human intervertebral disc. Spine (Phila Pa 1976) 1995;20:1307-14. [Crossref] [PubMed]
- Verbiest H.. Pathomorphologic aspects of developmental lumbar stenosis. Orthop Clin North Am 1975;6:177-96. [PubMed]
- Cheung JP, Cheung PW, Cheung KM, et al. Decompression without Fusion for Low-Grade Degenerative Spondylolisthesis. Asian Spine J 2016;10:75-84. [Crossref] [PubMed]
- Samartzis D, Cheung JP, Rajasekaran S, et al. Critical Values of Facet Joint Angulation and Tropism in the Development of Lumbar Degenerative Spondylolisthesis: An International, Large-Scale Multicenter Study by the AOSpine Asia Pacific Research Collaboration Consortium. Global Spine J 2016;6:414-21. [Crossref] [PubMed]
- Williams R, Cheung JP, Goss B, et al. An International Multicenter Study Assessing the Role of Ethnicity on Variation of Lumbar Facet Joint Orientation and the Occurrence of Degenerative Spondylolisthesis in Asia Pacific: A Study from the AOSpine Asia Pacific Research Collaboration Consortium. Global Spine J 2016;6:35-45. [Crossref] [PubMed]
- Malmivaara A, Slatis P, Heliovaara M, et al. Surgical or nonoperative treatment for lumbar spinal stenosis? A randomized controlled trial. Spine 2007;32:1-8. [Crossref] [PubMed]
- Weinstein JN, Tosteson TD, Lurie JD, et al. Surgical versus nonoperative treatment for lumbar spinal stenosis four-year results of the Spine Patient Outcomes Research Trial. Spine 2010;35:1329-38. [Crossref] [PubMed]
- Weinstein JN, Tosteson TD, Lurie JD, et al. Surgical versus nonsurgical therapy for lumbar spinal stenosis. N Engl J Med 2008;358:794-810. [Crossref] [PubMed]
- Ng KKM, Cheung JPY. Is minimally invasive surgery superior to open surgery for treatment of lumbar spinal stenosis? A systematic review. J Orthop Surg (Hong Kong) 2017;25:2309499017716254. [PubMed]
- Komp M, Hahn P, Oezdemir S, et al. Bilateral spinal decompression of lumbar central stenosis with the full-endoscopic interlaminar versus microsurgical laminotomy technique: a prospective, randomized, controlled study. Pain Physician 2015;18:61-70. [PubMed]
- Ruetten S, Komp M, Merk H, et al. Surgical treatment for lumbar lateral recess stenosis with the full-endoscopic interlaminar approach versus conventional microsurgical technique: a prospective, randomized, controlled study. J Neurosurg Spine 2009;10:476-85. [Crossref] [PubMed]
- Bloom BS. Use of formal benefit/cost evaluations in health system decision making. Am J Manag Care 2004;10:329-35. [PubMed]
- Lønne G, Johnsen LG, Aas E, et al. Comparing cost-effectiveness of X-Stop with minimally invasive decompression in lumbar spinal stenosis: a randomized controlled trial. Spine 2015;40:514-20. [Crossref] [PubMed]
- Parker SL, Anderson LH, Nelson T, et al. Cost-effectiveness of three treatment strategies for lumbar spinal stenosis: Conservative care, laminectomy, and the Superion interspinous spacer. Int J Spine Surg 2015;9:28. [Crossref] [PubMed]
- Hospital Authority. Hospital Authority Ordinance (Chapter 113): Revisions to List of Charges, G.N. 1488. 2013.
- Cheung PWH, Fong HK, Wong CS, et al. The influence of developmental spinal stenosis on the risk of re-operation on an adjacent segment after decompression-only surgery for lumbar spinal stenosis. Bone Joint J 2019;101-b:154-61.
- Wong CKH, Cheung JPY, Cheung PWH, et al. Traditional growing rod versus magnetically controlled growing rod for treatment of early onset scoliosis: Cost analysis from implantation till skeletal maturity. J Orthop Surg (Hong Kong) 2017;25:2309499017705022. [Crossref] [PubMed]
- National Institute for Health and Clinical Excellence (NICE). Guide to the methods of technology appraisal 2013: process and methods guides. London: NICE, 2013.


