Concomitant back and leg pain as predictors for trajectories of poor outcome after single level lumbar micro-decompression alone and with micro-discectomy: a study of 3,308 patients
Introduction
Lower-limb nerve root pain caused by a lumbar disc herniation is the most frequent indication for spinal surgery. Sixty-thousand such operations are performed in France each year (1). The US has also witnessed a marked increase (2.1 per 1,000 Medicare enrolees) in the rates of lumbar discectomies in the past decade (2). However, the literature reports that one in four patients has a suboptimal outcome following surgery (3), suggesting that pre-operative identification of risk factors associated with a poor outcome is paramount for appropriate case selection.
Lumbar spine stenosis (LSS) is the most common indications for lumbar spine surgery in people older than 65 years. Cumulative data reveal that patients with severe LSS associated with excruciating leg and back pain benefit from decompressive surgery (4). The primary goal of surgical intervention in LSS is to decompress the neural structures, focusing on relieving the leg symptoms and less on improving back pain. Therefore, although back pain does improve, the improvement in leg pain (LP) is usually greater. Patients with LSS and predominant LP have better surgical outcomes than patients with predominant back pain or equally bothersome pain in the legs and the back (5).
Surgical success depends on achieving pain relief, which improves quality of life (6). Despite many studies on the predictors of outcome (7-9), the effect of the proportion of pre-operative pain that is radicular (leg) compared to axial (back), has not been studied extensively. Though a few studies (6,10-12), indicate that greater preoperative low back pain (LBP) is associated with a poor outcome, none have investigated the extent of this relationship and whether the relative intensity of radicular and axial pain can be used to guide patient selection or aid patient counselling prior to surgery.
It is recognized that to identify and monitor the various factors that affect the course of a disease or a surgery outcome is of great importance to clinical practice. However, conventional statistical practice generally falls far short of taking full advantage of the information available in multivariate longitudinal data for tracking the course of the outcome of interest.
In recent years, group-based trajectory models (GBTMs) have been increasingly applied in clinical research to assess heterogeneity in response to clinical interventions (13).
GBTMs offers a number of applications in clinical research. This includes description of the course of an outcome over age or time, assessing the impact of therapeutic interventions, and identifying predictors of trajectory group membership.
The aims of this study are to model the Core Outcome Measures Index (COMI) score and pain visual analogue scales (VAS) trajectory-groups and to investigate the influence and predictive value of the relative intensity of axial and radicular pain on the outcome of lumbar decompressive and discectomy surgery. A secondary objective was to develop a classification tree to identify the most likely outcome route for each patient after the intervention that might help to manage patients’ expectations.
Methods
All patients in our centre were registered on a prospective basis in an in-house database linked to Euro-spine’s Spine Tango Spine Surgery Registry (14), allowing documentation of baseline pathology, medical history, surgical procedures and complications, as well as patient-rated outcome at baseline and repeated follow-up.
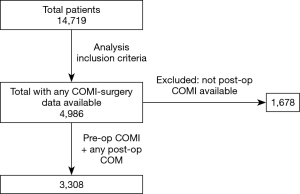
Consecutive patients who underwent single level lumbar micro-decompression and or micro-discectomy performed at a single tertiary level spinal centre between August 2011 and September 2017 were included in this study. Written informed consent for data collection was obtained from all patients. Patients with a clinical and confirmed radiological diagnosis of degenerative lumbar spine disorders producing radicular pain (lumbar disc herniation, degenerative stenosis and degenerative spondylolisthesis) were selected for surgery. Patients who underwent fusion surgery, surgery at more than one level, motion preserving stabilization, were excluded. Patients without pre-operative and at least one post-operative COMI questionnaire available were excluded from this analysis.
For each case the operating surgeon completed the SSE Spine Tango 2011 surgery form (15) or SSE Spine Tango 2017 surgery form, which included the diagnosis, clinical data and details of surgery. The COMI questionnaire (16) was used to assess outcome. The pain questions in the COMI questionnaire recorded the intensity of LBP (LBP VAS) and LP (LP VAS) separately on a 0–10 VAS scale, with 0 indicating no pain and 10 indicating the worst imaginable pain. Patients completed a (COMI) questionnaire pre-operatively and at 3 months (postal questionnaire). At follow-up the additional COMI questions included the occurrence and nature of any complications that may have risen following the surgery, any reoperations (at the same or different levels), satisfaction with the result of treatment and finally, the global outcome of surgery. The COMI score was calculated using standard means (16).
Smoking status and previous diagnosis of anxiety or depression were obtained from electronic patient record (EPR).
Based on the response to the patient reported global outcome question “Overall, how much did the treatment that you received (the operation) help your back problem?”, the patients were dichotomised into two outcome groups - good outcome (response 1—“helped a lot”, and 2—“operation helped”) and poor outcome (response 3—“operation helped a little”, 4—“operation did not help”, and 5—“operation made things worse”).
Ethical statement
NHS Rec approval not required for this service evaluation. Written consent gained from all patients to use collected data.
Surgical technique
All surgeries were performed through a posterior midline incision. A lumbar decompression in the form of a discectomy, sequestrectomy, hemilaminectomy, facet joint resection (partial), partial laminectomy, foraminotomy, or laminotomy of the lumbar spine was performed under magnification, using an operating microscope.
Statistical analyses
Descriptive pre-operative data are shown as mean and standard deviation (SD). Categorical variables are expressed as frequencies and percentages. Comparison between the patients included versus patients excluded in the analysis was performed using t-test for the normally distributed continuous variables, Wilcoxon rank-test for the non-normally distributed continuous variables and chi-squared test for the categorical variables.
Our population was divided in two groups: patients with discectomy and patients with decompression alone. The analysis were performed independently in both groups and also combined as a heterogeneous group of patients.
Post-operative data and mean changes from pre-operative to post-operative were presented. Also, mean pre-operative COMI score, LBP VAS and LP VAS, difference between LBP VAS and LP VAS (LBP–LP) and demographics were by presented by outcome group (poor/good). Means and percentages were compared using Wilcoxon rank-test and chi-squared test.
Minimum clinically relevant change (MCRC) in COMI score, LBP VAS and LP VAS was calculated as an improvement of at least 2.2 points from pre to post-operative COMI questionnaire (17) and percentages of patients achieving MCRC were plotted.
Patients with pre-operative COMI and at least one post-operative follow-up COMI were included in the analysis. For this analysis we used a two-step approach. First, COMI score, LBP VAS and LP VAS trajectories of patients were modelled using a form of discrete mixture-modelling called group-based trajectory analysis (GBTM) (18). GBTM is a statistical method that is designed to identify a finite number of groups of individuals following similar trajectories over age or time of multiple outcomes. It assigns each individual to a trajectory group, based on Bayesian posterior probabilities for group membership. Group-based trajectories were modelled in groups ranging from two to four trajectories. The optimal number of possible trajectories was determined by statistical fit and the clinical relevance.
In the second step, multivariable multinomial logistic regression model was used to find possible predictors of membership to each trajectory group. Predictors included pre-operative measures of COMI score, LBP VAS, LP VAS, LBP–LP, morbidity [American Society of Anaesthesiologists (ASA) class], type of degeneration (disc prolapse, Lumbar canal stenosis, Spinal deformity/instability, none), smoking status, level of intervention (L1–L2, L2–L3, L3–L4, L4–L5 and L5–S1), previous surgery at the same level, re-interventions after index surgery, symptoms duration before surgery, previous diagnosis of anxiety or depression, BMI, gender, age and the answers to the question: “Which of the following problems troubles you the most?” (1. back pain, 2. leg/buttock pain (sciatica), 3. sensory disturbances in the back/leg/buttocks or none of these). Factors statistically significant in univariate analysis were included in the multivariable model.
We performed classification tree analysis (19) using the recursive partitioning method using rpart R package, pruned to a complexity parameter of 0.005 with 10-fold cross-validation.
Data analysis was conducted using STATA V.14.0, user-written STATA TRAJ (20) and R version 3.5.1.
Results
Pre-operative characteristics
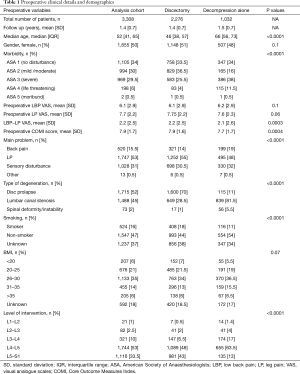
After exclusion using the criteria described in the methods, 3308 patients were available for this analysis (Figure 1). All had undergone single level primary micro-decompressive surgery by 39 senior orthopaedic/neuro-spinal surgeons at a single tertiary centre. The mean follow-up was 1.4 years (SD: 0.7), median age of 52 years [interquartile range (IQR): 41, 65] and 50% female. Before the intervention, 34% of patients presented ASA 1, 30% ASA 2, 29.5% ASA 3, 6% ASA 4, and 0.5% ASA 5. Mean LBP VAS 6.1 (SD: 2.9), mean LP VAS 7.7 (SD: 2.2) and mean COMI score 7.9 (SD: 1.7) (Table 1). Sixteen percent of patients were smokers. The majority of interventions were located in L4–L5 (53%) and L5–S1 (33.5%) segments. Patients with decompression alone were older than patients with discectomy. They also presented more severe morbidity (ASA 3 & ASA 4), a smaller proportion of smokers and more reported back pain as the most troublesome problem when compared with discectomy patients.
Full table
COMI and pain VAS changes—MCRC
Mean COMI score, LBP VAS and LP VAS were significantly reduced after the intervention with an overall mean change of 3.5, 2.1 and 4 points respectively (Table 2).

Full table
Sixty-three percent of patients reached COMI score MCRC defined as an improvement of at least 2.2 points, 42% reached LBP VAS MCRC and 62% LP VAS MCRC (Figure 2).

Patient reported global outcome
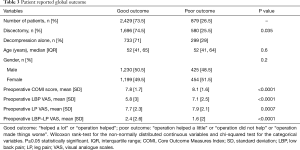
More than 73% of patients reported good outcome after surgery (“helped a lot” or “operation helped”). These patients showed statistically significant lower mean pre-operative values for COMI score (7.8, SD: 1.7), LBP VAS (5.8, SD: 3) and LP VAS (7.7, SD: 2.3) than the patients reporting poor outcome (8.1, SD: 1.6; 7.1, SD: 2.5; 7.9, SD: 2.1; respectively). Also, the difference between pre-operative LBP VAS and LP VAS was significantly larger in those patients reporting a good outcome (Table 3). Twenty-nine percent of patients with decompression alone reported poor outcome; 25.5% patients with discectomy also reported poor outcome.
Full table
Group-based multi-trajectory model
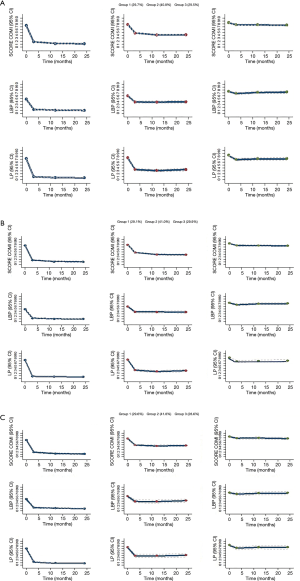
The group-based multi-trajectory model was selected by statistical fit, using the Bayesian Information Criterion (BIC), and the clinical relevance. Overall, we identified three trajectory-groups or clusters of individuals following similar trajectories across COMI score, LBP VAS and LP VAS. Almost 90% of patients had a >0.75 probability of membership in their assigned multi-trajectory group.
First, a trajectory-group of patients with large-improvement (LI) during the follow-up time with 29.7% of the patients; second, a trajectory-group with moderate-improvement (MI) encompassing 40.8% of the patients and third, a group showing no-improvement (NI) including 29.5% of the patients (Figure 3A).
Discectomy and decompression alone groups
The independent trajectory analyses for the patients who also underwent discectomy (Figure 3B) and for the group with decompression alone (Figure 3C) were practically identical to the overall analysis, with similar trajectory-groups sizes and scores improvements post-operative, also showing the same three trajectory-groups LI, MI and NI described in the overall population.
When the preoperative characteristics of these three overall trajectory-groups were compared we found significant differences in COMI scores, LBP VAS, LP VAS, morbidity, type of degeneration, smoking status and main problem reported (Table 4). NI group showed the highest values for COMI score VAS (8.5, SD: 1.25), LBP VAS (7.6, SD: 2.2) and LP VAS (8.5, SD: 1.7) and a larger proportion of smokers (25%) compared with the other two groups (P<0.0001). NI also presented slightly larger proportion of patients with more severe morbidity (ASA 3 and ASA 4) than the other two groups of patients. The LI group had the lowest preoperative LBP VAS score. In addition, the mean difference between axial and radicular pain VAS scores was the largest in the LI group (2.4) and the smallest in the NI group (0.8). NI also showed significantly higher percentages of back pain (22%) and sensory disturbance (34.5%) and lower proportion of LP (43%), as the main problem reported by the patients, when compared with LI (10%, 26.5%, 63%) and MI (15%, 32%, 52.5%).
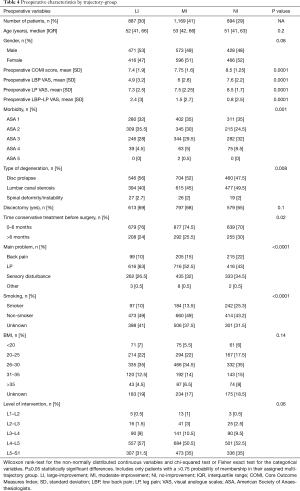
Full table
MCRC and patient reported global outcome by trajectory group
Almost 100% of the patients from LI group and nearly 85% from MI group reported good global outcome from surgery. Over 68% of the patients from the NI group reported poor global outcome (Table S1).
When we look at the MCRC achieved by trajectory-group we observed that 96%, 64% and 91% achieved a clinically relevant change in COMI score, LBP and LP respectively in the LI group. In the other hand, NI group patients only achieved it on 18%, 15% and 21% respectively (Figure S1).
Multinomial logistic regression
After multinomial logistic regression analysis in the overall population, the factors associated with NI when compared with LI were higher preoperative LBP VAS, LP VAS and COMI score, to be smoker, presence any of post-operative surgical complications before discharge, previous surgery at the same level, to have been on conservative treatment for more than 6 months previous to surgery and to have had previous diagnosis of anxiety or depression (Table 5). Variables associated with MI trajectory when compared to LI were higher preoperative LBP VAS and COMI scores, to be smoker and being female. In the discectomy group, the factors associated with NI when compared with LI were higher preoperative LBP VAS and COMI score, to be smoker, previous surgery at the same level and being female. In the group of patients with decompression alone the factors associated with NI when compared with LI were higher preoperative LBP VAS and COMI score, to be smoker, presence any of post-operative surgical complications before discharge and previous surgery at the same level.
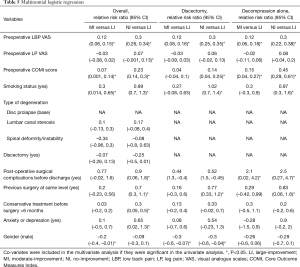
Full table
Classification tree
The classification tree (Figure 4) shows that LBP is the most important factor to split the patients as NI or LI. Also, it identified global COMI score, smoking status and previous surgery at the same level as influential predictors of NI group. Overall, the tree offers a classification accuracy of 75% (CI: 70–79%) in the validation sample. A LBP ≥6.5 and global COMI score ≥7.1 indicates NI membership; also, previous surgery at the same level and smoking appears as important factors to split the patients between LI and NI groups.
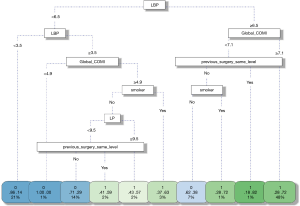
Discussion
This represents the largest study to investigate the influence and predictive value of the relative intensity of axial and radicular pain on the outcome of lumbar micro-decompressive, for both discectomy and decompression alone, surgery. It is also the first study to define trajectory-groups of patients using GBTM in the setting of surgical decompression in the management of radicular pain secondary to lumbar degenerative disease. In addition, this study is the first one to use classification and regression tree (CART) analysis to classify patients’ outcome after micro-decompression surgery based on the pre-operative features.
We identified three well-delimited and robust groups of patients based on the joint progression of global COMI score, LBP VAS and LP VAS during a period of 2 years after surgery. Although a majority of patients presented high preoperative values for COMI score and both LBP and LP scales, more than 70% showed a quick improvement in the first 3 months after surgery, no matter whether they had discectomy or decompression alone. This is in line with previous reports (3). On the other hand, we report that almost a third of patients showed a maintained high global COMI score and high back and LP during that time in the NI trajectory-group. We found that higher preoperative LBP VAS strongly predicts membership for the NI group. Similar findings have been described previously (3,8,11,12,21); in addition, our study shows that these patients in the NI group had also very high levels of LP and equally high global COMI score and that these patients were not improving significantly in any of the three scores 2 years after the surgical intervention. In this group, only 18%, 21% and 15% reached MCRC for COMI score, LP and LBP respectively and the difference between preoperative LBP and LP was significantly smaller (mean: 0.8) than in LI and MI with mean differences of 2.4 and 1.5. This difference between back and LP was due mostly to the intensity of back pain, confirming that the preoperative LBP plays a major role in the outcome after surgery. Kleinstueck et al. (12) reported that patients with more back pain showed significantly worse outcomes after decompression surgery for lumbar herniation disc and that LP intensity did not differ significantly between the outcome groups, concluding that it was only the level of LBP, not LP, that was predictive of outcome and that LBP and LP need to be distinguished as independent predictors in their own right.
Our analysis allowed us to obtain a deeper understanding of the influence of pre-operative LBP and LP on the surgery outcome. The MI trajectory is characterised by a moderate preoperative LBP and in general more moderate preoperative symptoms than NI, with partial reduction in the global COMI score and pain during the follow up time, while LI shows low LBP and a massive decrease of these scores. This gives us the possibility not only to distinguish between patients with merely good or poor outcome, but also to distinguish those patients who will obtain only partial benefits from the intervention. This more accurate information might help to manage patients’ expectations in a more precise way.
The fact that discectomy and decompression alone gave us similar trajectories after surgery reinforces the importance of preoperative LBP and LP as predictors for both types of intervention.
A link between smoking and a poor spinal surgery outcome has been previously proposed (22,23). Chronic smoking has significant adverse effects on the human spinal column. It has been shown that nicotine down-regulates both the proliferation rate and biosynthetic activity of disc cells (24). In addition, smoking causes the constriction of the vascular network surrounding the intervertebral discs. An interesting study in 825 patients reported that non-smokers experienced a significantly LI at 1 year following micro-decompression for LSS compared to smokers and that smokers were less likely to achieve a minimal clinically important difference. However, the authors highlighted that, even if significantly smaller than non-smokers, considerable improvement also was found among smokers (22). Nerland et al. investigated the frequency and predictors of deterioration after decompressive surgery for single and two-level lumbar spinal stenosis in 1,735 patients. Their findings confirmed smoking as a significant predictor of deterioration in functional status postoperatively (23).
In line with these findings, our study showed that to be a smoker was one of the strongest factors increasing the risk of a worse outcome trajectory for patients with decompression alone and for patients with discectomy, NI, with an increased risk to follow MI trajectory-group for patients that otherwise might have followed LI trajectory, and almost double chances to be in NI than in LI group. Further research on smoking would be necessary to accurately estimate its effects on surgery outcome assessing the dosages, frequency and current or former smoking status and also stratifying the patients by type of intervention.
Anxiety, depression and others psychological factors have been reported to affect outcome after lumbar spine surgery (25-28). In a recent comparative analysis of the quality of life and functionality of patients undergoing lumbar spine arthrodesis due to degenerative lumbar spine disease was found that patients without anxiety and depression symptoms had better results on the scales compared to those with such symptoms (27). Moreover, a systematic review on predictors of outcomes following lumbar surgery concluded that self-reported levels of depression, anxiety, coping, somatization and hypochondriasis were found to be associated with greater risk for poor outcome in most studies (25). Therefore, it is not completely unexpected that our study shows similar findings. In this case, we found that patients who have been previously diagnosed with anxiety or depression strongly predicted membership to the NI outcome trajectory-group but was not predictive of MI. It would be worthy to consider the recommendation of assessment for anxiety or depression in patients undergoing lumbar decompression surgery and offer information and preparation leading up to surgery to address underlying psychological factors.
CART analysis has been previously use to generate personalised information to help patients make informed decisions (29). However, it has never been use in lumbar micro-decompressive surgery. The CART (19) offers a clearer interpretation of the factors linked to NI or LI, and in addition allows predicting the patient’s trajectory-group membership with 75% accuracy. Larger study population and bigger validation samples might lead to a higher classification success rate. This model could be helpful for the clinician in order to manage expectations and inform the patients prior the intervention, and could be even used as a possible decision-making tool, it is intuitive and it has demonstrated a positive prediction of surgery outcomes.
The major strengths of our analysis is the large sample size, the routine clinical settings from which the data were extracted and a follow-up up to 2 years after surgery. We were able to adjust our analysis for the most relevant co-variables including important factors such as smoking status, BMI and psychological factors which were included in the surgery forms in 2011.
Our study has some limitations. The inclusion of different degenerative pathologies could lead to some heterogeneity and, therefore, some bias. However, we performed parallel analysis for discectomy and for decompression alone to account for these differences and, in addition, we allowed for type of degeneration and whether the patient had discectomy performed in the overall multinomial logistic regression model to take into account that heterogeneity.
Another limitation may be the lack of detail on the smoking status, with only current smoker/non-smoker information available. It would be desirable to add frequency and amount of smoking and also ex-smokers data such as time without smoking before the intervention.
Our study, presents limitations common to all studies within a real-world clinical setting, such as patients loss to follow up and missing data. Those patients lost to follow up were excluded of the analysis. Patients with sufficient data available were representative of the overall cohort.
Conclusions
This study is the first to successfully clustered patients that underwent after Single Level Lumbar Micro-Decompression Surgery into three clear trajectories of outcome after surgery. We concluded that preoperative LBP is a key factor for surgery outcome for both type of interventions, discectomy and decompression alone, but cannot be considered independently but in combination with other factors such as smoking, psychological factors, duration of conservative treatment and existence of previous surgeries. This is very useful while counselling and managing patients’ expectations, helping patients to make better decisions, generating more realistic expectations about surgery outcomes, and possibly increasing satisfaction with treatment.
Acknowledgments
Funding: None.
Footnote
Data Sharing Statement: Available at http://dx.doi.org/10.21037/jss-19-462
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/jss-19-462). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). NHS Rec approval not required for this service evaluation. Written consent gained from all patients to use collected data.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Blamoutier A.. Surgical discectomy for lumbar disc herniation: surgical techniques. Orthop Traumatol Surg Res 2013;99:S187-96. [Crossref] [PubMed]
- Weinstein JN, Lurie JD, Olson PR, et al. United States' trends and regional variations in lumbar spine surgery: 1992-2003. Spine (Phila Pa 1976) 2006;31:2707-14. [Crossref] [PubMed]
- Parker SL, Mendenhall SK, Godil SS, et al. Incidence of low back pain after lumbar discectomy for herniated disc and its effect on patient-reported outcomes. Clin Orthop Relat Res 2015;473:1988-99. [Crossref] [PubMed]
- Slätis P, Malmivaara A, Heliövaara M, et al. Long-term results of surgery for lumbar spinal stenosis: a randomised controlled trial. Eur Spine J 2011;20:1174-81. [Crossref] [PubMed]
- Lurie J, Tomkins-Lane C. Management of lumbar spinal stenosis. BMJ 2016;352:h6234. [Crossref] [PubMed]
- Jansson KA, Nemeth G, Granath F, et al. Health-related quality of life in patients before and after surgery for a herniated lumbar disc. J Bone Joint Surg Br 2005;87:959-64. [Crossref] [PubMed]
- den Boer JJ, Oostendorp RA, Beems T, et al. A systematic review of bio-psychosocial risk factors for an unfavourable outcome after lumbar disc surgery. Eur Spine J 2006;15:527-36. [Crossref] [PubMed]
- Kerr D, Zhao W, Lurie JD. What are long-term predictors of outcomes for lumbar disc herniation? A randomized and observational study. Clin Orthop Relat Res 2015;473:1920-30. [Crossref] [PubMed]
- Lurie JD, Tosteson TD, Tosteson A, et al. Long-term outcomes of lumbar spinal stenosis: eight-year results of the Spine Patient Outcomes Research Trial (SPORT). Spine (Phila Pa 1976) 2015;40:63-76. [Crossref] [PubMed]
- Haugen AJ, Brox JI, Grovle L, et al. Prognostic factors for non-success in patients with sciatica and disc herniation. BMC Musculoskelet Disord 2012;13:183. [Crossref] [PubMed]
- Hill JC, Konstantinou K, Egbewale BE, et al. Clinical outcomes among low back pain consulters with referred leg pain in primary care. Spine (Phila Pa 1976) 2011;36:2168-75. [Crossref] [PubMed]
- Kleinstueck FS, Fekete T, Jeszenszky D, et al. The outcome of decompression surgery for lumbar herniated disc is influenced by the level of concomitant preoperative low back pain. Eur Spine J 2011;20:1166-73. [Crossref] [PubMed]
- Nagin DS, Odgers CL. Group-based trajectory modeling in clinical research. Annu Rev Clin Psychol 2010;6:109-38. [Crossref] [PubMed]
- EUROSPINE. 2016. [cited 2016 Jan 1]. Available online: http://www.eurospine.org/spine-tango.htm
- Röder C, El-Kerdi A, Grob D, et al. A European spine registry. Eur Spine J 2002;11:303-7. [Crossref] [PubMed]
- Mannion AF, Elfering A, Staerkle R, et al. Outcome assessment in low back pain: how low can you go? Eur Spine J 2005;14:1014-26. [Crossref] [PubMed]
- Munting E, Roder C, Sobottke R, et al. Patient outcomes after laminotomy, hemilaminectomy, laminectomy and laminectomy with instrumented fusion for spinal canal stenosis: a propensity score-based study from the Spine Tango registry. Eur Spine J 2015;24:358-68. [Crossref] [PubMed]
- Nagin DS, Jones BL, Passos VL, et al. Group-based multi-trajectory modeling. Stat Methods Med Res 2018;27:2015-23. [Crossref] [PubMed]
- Speybroeck N.. Classification and regression trees. Int J Public Health 2012;57:243-6. [Crossref] [PubMed]
- Jones BL, Nagin DS. A note on a Stata plugin for estimating group-based trajectory models. Sociol Methods Res 2013;42:608-13. [Crossref]
- Jansson KA, Nemeth G, Granath F, et al. Health-related quality of life (EQ-5D) before and one year after surgery for lumbar spinal stenosis. J Bone Joint Surg Br 2009;91:210-6. [Crossref] [PubMed]
- Gulati S, Nordseth T, Nerland US, et al. Does daily tobacco smoking affect outcomes after microdecompression for degenerative central lumbar spinal stenosis? - A multicenter observational registry-based study. Acta Neurochir (Wien) 2015;157:1157-64. [Crossref] [PubMed]
- Nerland US, Jakola AS, Giannadakis C, et al. The risk of getting worse: predictors of deterioration after decompressive surgery for lumbar spinal stenosis: a multicenter observational study. World Neurosurg 2015;84:1095-102. [Crossref] [PubMed]
- Elmasry S, Asfour S, de Rivero Vaccari JP, et al. Effects of tobacco smoking on the degeneration of the intervertebral disc: a finite element study. PLoS One 2015;10:e0136137. [Crossref] [PubMed]
- Celestin J, Edwards RR, Jamison RN. Pretreatment psychosocial variables as predictors of outcomes following lumbar surgery and spinal cord stimulation: a systematic review and literature synthesis. Pain Med 2009;10:639-53. [Crossref] [PubMed]
- Chapin L, Ward K, Ryken T. Preoperative depression, smoking, and employment status are significant factors in patient satisfaction after lumbar spine surgery. Clin Spine Surg 2017;30:E725-32. [Crossref] [PubMed]
- Netto MB, Barranco ABS, Oliveira KWK, et al. Influence of anxiety and depression symptoms on the quality of life in patients undergoing lumbar spine surgery. Rev Bras Ortop 2017;53:38-44. [PubMed]
- McKillop AB, Carroll LJ, Battie MC. Depression as a prognostic factor of lumbar spinal stenosis: a systematic review. Spine J 2014;14:837-46. [Crossref] [PubMed]
- Gutacker N, Street A.. Use of large-scale HRQoL datasets to generate individualised predictions and inform patients about the likely benefit of surgery. Qual Life Res 2017;26:2497-505. [Crossref] [PubMed]