
Full endoscopic interlaminar discectomy (FEID) for recurrent lumbar disc herniation: surgical technique, clinical outcome, and prognostic factors
Introduction
Recurrent lumbar disc herniation (RLDH) is defined as the occurrence of herniated disc material at the same level in a patient who has undergone discectomy. The rate of recurrence of disc herniation reported in the literature varies from 5% to 18% (1-3). Young patients with high disability and without a neurological deficit are at the highest risk of recurrent disc herniation (4). Advanced disc degeneration and decreased disc height were also noted to be positively correlated with recurrence (5). Comparing the initial technique of discectomy, there is no significant difference in recurrent rates after microdiscectomy or sequestrectomy, but long-term functional outcome after sequestrectomy is superior at 2 years (6-8). Nonoperative treatment is considered in the symptomatic patients without neurologic deficit; however, patients undergoing revision surgery for RLDH improved significantly compared to baseline (4). Operative treatment is indicated for those patients with neurologic deficits or symptoms refractory to conservative measures. The main options of surgical treatment for RLDH consist of revision lumbar discectomy and instrumented fusion. Clinical outcomes and complication rates were similar between the two treatments in patients without radiographic instability. However, patients with repeated discectomy had shorter operative times, length of stay, quicker recovery and lower financial costs as compared to patients undergoing instrumented fusion (9). Open discectomy has been the standard treatment of RLDH when there is no pre-existing segmental instability, spinal deformity, or chronic low back pain (10). Revision discectomy is challenging because of the adhesion may made the dissection plane between neural and scar tissue difficult to be recognized and wide exploration to posterior structures may lead to segmental instability (11,12). This difficulty may cause additional problems such as bleeding, dural tear, nerve injury and inadequate decompression (13). Endoscopic techniques have been widely applied in the treatment of disc herniation (14-16). Two approaches can be used for full endoscopic lumbar discectomy: the transforaminal or the interlaminar approach (17). Transforaminal approach is introduced through the neural foramen and can be performed at all lumbar levels except for L5-S1 in patients with high iliac crests (18,19). The route of this approach usually is not involved in the previous open surgery and hence the scar tissue can be avoided in revision cases (14,15,20). The interlaminar approach is reserved for L5-S1 or L4-5 levels where the interlaminar space is sufficient or when transforaminal approach is not feasible. The transforaminal route may be blocked by the iliac crest, a large L5 transverse process, a large facet joint, narrowed disc space or previously implanted instruments. The minimally invasive nature of endoscopic interlaminar technique including preservation of dorsal musculature and posterior elements may reduce the risk of segmental instability after revision surgery (21). Some studies showed that the problems related to adhesive scar tissues or postoperative instability encountered by open interlaminar approach could be overcome by endoscopic methods (15,22). The object of this study is to present the surgical technique and outcomes of FEID for recurrent disc herniation.
Methods
The trial was conducted in accordance with the Declaration of Helsinki and the Harmonized Tripartite Guideline for Good Clinical Practice from the International Conference on Harmonization. This study was reviewed by the Institutional Review Board (IRB) of Buddhist Dalin Tzu-Chi General Hospital Foundation (IRB number: B10901026) and written informed consent was obtained from all patients.
Patients
From June 2014 to December 2016, full endoscopic interlaminar discectomy (FEID) was performed in consecutive 24 patients for RLDH at the same level and ipsilateral side after conventional open discectomy. All patients had an improved pain interval for at least 3 months after primary discectomy. The inclusion criteria involved: (I) recurrent leg radiation pain that failed at least 3 months of conservative treatment excluding severe intractable pain or accompanying motor weakness, (II) RLDH was confirmed by magnetic resonance imaging (MRI), and (III) MRI findings were correlated with clinical symptoms and sings. The exclusion criteria were: (I) segmental instability due to spondylolisthesis, facet joint arthritis or wide decompression performed in initial surgery, (II) spinal infection, tumor or fracture, (III) coexisting psychological diseases. There are 10 female and 14 male patients with L4/5 being involved in 8 cases and L5/S1 in 16 cases. Their age ranged from 25 to 60 years (mean 44.6 years).
Surgical technique
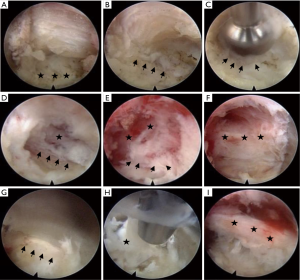
The surgical procedure was performed in prone position and under general anesthesia. The position of entry point for the recurrent disc herniation was determined under fluoroscopic guidance and, for the safety, close to the lateral margin of the interlaminar window (Figure 1A). After a small skin and fascia incision (8 mm), a dilator (6.9 mm) was introduced and docked to the lateral bony edge of interlaminar window to avoid direct penetration to the epidural space. The position of interlaminar space could be determined by palpating the surrounding bony structure with the dilator. A working sheath with a beveled opening (8.0 mm outer diameter) was introduced through the dilator and the final position was verified by the fluoroscope (Figure 1B). The surgery was performed after introducing an endoscope (SPINENDOS GmbH, Germany) to remove soft tissues including residual muscle connecting and scar tissues until the bony margin of the medial facet joint and lamina was visualized (Figure 2A,B). Further laminectomy might not be needed because sufficient laminar window already had been created at previous operation. However, in the condition of adhesion or severe stenosis, part of the osseous margin including lamina or facet joints (about 2–4 mm) might have to be removed by using of high-speed drill (SPINENDOS GmbH, Germany) to separate scar tissue and to create an adequate working space (Figure 2C). All the soft tissues were separated from the medial facet joint with the dissector or curette after repeated gentle twisting motion under direct vision. The direction of dissection was from medial to lateral along the medial bony margin and it was important to feel the edge of the facet joint with the tip of the dissector during advancement (Figure 2D). The scar and soft tissues were detached from osseous structures (Figure 2E) and resected with a punch (Figure 2F). Ligamentum flavum (LF) might be noted after dissection of the soft tissue and it could be used as an anatomic landmark (Figure 2G). The LF was transgressed carefully to enter the epidural space (Figure 2H). The lateral margin of nerve root was recognized (Figure 2I). In severe adhesive cases, aggressive dissection of the scar from neural tissue was not attempted to avoid dural tear (Figure 2J). By using of a probe to retract the neural structure medially, the working sheath was rotated clockwise or counterclockwise and pivoted medially to further separate the scar tissue from the neural tissue. The scar tissue, nerve root and thecal sac were retracted together with the working sheath to avoid dural tear (Figure 2K). The herniated disc was removed through the opening in the beveled side of the working sheath (Figure 2L). If the herniated disc material was too large, the graspable part was removed first to reduce the tension of neural structures and create more working space (Figure 2M). The remained herniated material was removed with medial advancement and gradual rotation of the working sheath until all neural structures were retracted medially and only the herniated disc was in the view of the endoscope (Figure 2N). The annular defect could be identified by visualization or palpation, and all graspable disc material was removed through the defect. Decompression was confirmed after thorough examination of the dura sac and nerve root to make sure free movement of the neural tissue (Figure 2O). Finally, the working sheath and the endoscope were withdrawn and the skin was closed with 3-0 nylon.


Outcome assessment and statistical analysis
Outcomes were evaluated using the visual analog scale (VAS, 0–10) scores for back pain and Oswestry disability index (ODI, 0–100) scores for functional disability. The modified MacNab criteria were used for clinical global outcome assessment. Patients were asked to complete these questionnaires at pre-operation, and 2 weeks, 1 month, 3 months, 6 months, 12 months, and 24 months postoperatively. Preoperative and postoperative scores on the VAS, ODI, and modified MacNab criteria were compared using the Wilcoxon signed-rank test. Results were considered to be statistically significant if the P value was less than .05. Statistical analysis was performed using SPSS Version 13.0 (SPSS Inc., Chicago, IL, USA).
Results
General information
Twenty-four patients with 10 (41.7%) female and 14 (58.3%) male were enrolled in the present study. The levels operated were L4-L5 in 8 cases (33.3%) and L5-S1 in 16 cases (66.7%). Patient age averaged 44.6±8.2 years (range, 25–60 years) at surgery. The primary surgery was performed 78.2±32.3 months before the present FEID (range, 6–120 months). Comorbidity included 4 (16.7%) hypertension and 2 (8.3%) diabetes. The averaged skin-to-skin duration was 72.4 minutes (range, 45–125 minutes), and blood loss was minimal. On average, the hospital stay was 1.3 days after surgery (range, 1–5 days). In all cases, there were 2 severe adhesions between the scar and the neural tissues. Two patients encountered small dural tear which was detected at the end of procedure. Repair of the dural tear was not performed and no visible postoperative cerebrospinal fluid (CSF) leakage was detected. Both patients presented with mild transient neuropathy and recovered at 6 months follow-up. There was no major complication such as wound infection or severe neural injury.
Patient follow-up
Of the total 24 patients, re-recurrent disc herniation was noted within 3 months in 1 patient. The patient underwent repeated FEID and symptoms improved. One 58-year-old female patient underwent L4–5 fusion surgery 6 months after endoscopic revision discectomy due to back pain. VAS scores, ODI scores, and Modified MacNab Criteria were available for all patients preoperatively and at least 24 months follow-up. VAS and ODI scores significantly improved from preoperative baseline to the times at 1, 3, 6, 12 and ≥24 months post-surgery. The mean VAS of the trunk was 1.7±0.66, mean VAS of the limb was 1.5±0.78, and the mean ODI score was 8.2±5.14 at the last visit (Figures 3-5).
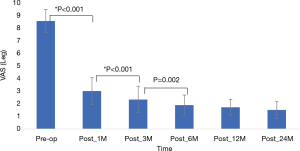
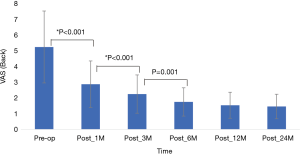
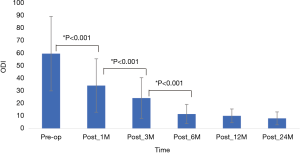
Modified MacNab criteria were used to assess the overall outcomes and the successful rate (excellent and good) was 87.5% (21/24) at one-year and 91.7% (22/24) at two-year follow-up. The patients who underwent additional operation were rated poor. Outcome results are summarized in Table 1. Among 24 patients, 10 who younger than 50 years old had 100% excellent results at post-operative 1 year and 2 years follow-up (Table 2).

Full table

Full table
Cases report
Case 1
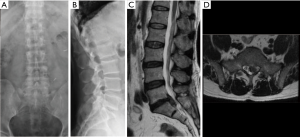
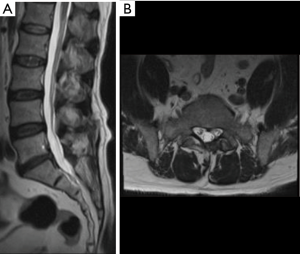
A 60-year-old male patient who had undergone open discectomy 10 years ago presented with left leg pain for 3 months. The VAS was 5/10 on the trunk and 9/10 on the left leg, and ODI was 65%. T2-weighted sagittal and axial MR imaging revealed a ruptured disc at L5–S1 on the left paramedian with downward migration. X-rays AP and lateral views showed the high iliac crest and large L5 transverse process (Figure 6). Left L5–S1 FEID via shoulder approach was selected. The target area was made on inferolateral corner of interlaminar window to remove the large downward herniated disc material. Postoperative T2-weighted MR imaging showed complete removal of the herniated disc material with an expanded thecal sac (Figure 7). After the operation, the patient’s symptoms improved and he was discharged from the hospital the day after surgery. At the 12-month postoperative follow-up, the VAS score was 2/10 on the trunk and 2/10 on the leg and the ODI was 15%.
Case 2
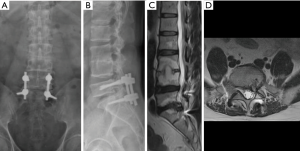
A 53-year-old male patient who had undergone L5-S1 discectomy and posterolateral fusion for 6 years presented with left leg pain and weakness for 2 months. X-rays AP (Figure 8A) and lateral views (Figure 8B) showed posterior decompression and instrumentation had been performed at L5-S1 level. T2-weighted sagittal (Figure 8C) and axial MR images (Figure 8D) revealed recurrent disc herniation at L5-S1. Considering the access of transforaminal approach might be blocked by the iliac crest and instrument, interlaminar approach was chosen. The procedure of endoscopic interlaminar discectomy was shown in Figure 9. The patient recovered well and was discharged the day after surgery.
Discussion
Open discectomy is the surgical treatment of choice for RHLD without objective evidence of spinal instability or stenosis (12,13,23). Scar tissue is the major concern which makes the revision surgery more difficult and increases the risk of dural tear or nerve injury. Further traumatization to posterior structures could lead to consecutive sequelae such as segmental instability (16). The satisfactory results of revision open discectomy after traditional open discectomy are about 70% (23). The results are rated worse when compared to primary discectomy, which is due to epidural scarring, progressive degeneration with stenosis, arachnoiditis, segmental instability or further injury to the posterior spinal structure (15,23). The complication rate after repeated open discectomy is reported as 13%, dural tears or infections being the most common problems (2). To improve outcome, tissue-sparing techniques are used for RLDH and technical advances have enabled a full-endoscopic procedure in revision cases. With full-endoscopic discectomy by transforaminal or interlaminar approach, excellent or good outcomes were achieved in about 90% of patients and the clinical results were reported as equal to that of the microsurgical technique (15). Compared to open surgery, a shorter operation time and fewer complications such as dural injury, dysesthesia, urinary retention, delayed wound healing, and infection were noted after endoscopic surgery (15).
By using of transforaminal approach, the spinal canal can be reached more safely in revision cases but adequate resection of the herniated discs is not always possible. The working mobility can be limited by a narrow neural foramen and the exiting nerve root, and the access may be disturbed by the iliac crest or a large transverse process (15). The interlaminar approach should be chosen when transforaminal route is not feasible; however, scar tissue is a troublesome problem as it is in open surgery. During FEID, the surgical principles are similar to that of open discectomy. For a safe approach, the osseous margin is first identified under direct vision. Different techniques related to dissection of scar tissue have been reported: (I) with the working channel to dissect scar tissue from the bony margin (15), (II) with a sharp freer, an endoscopic curette, and a specially designed micro-osteotome (16), or (III) the scar tissue is not dissected from the neural tissue, but is dissected from the medial facet joint, and the scar and the neural tissues were retracted together to avoid the problem of dural tear (22). In the present study, we use a dissector or curette to separate scar tissue from bony edge and additional resection of lamina or facet with high speed drill is performed in adhesive cases. It is relatively safe to dissect scar tissue from the bone. The area lateral to the laminar window usually is not involved in the primary surgery, and a plane between epidural scar tissue and herniated disc fragment can be found after a little bone edge (2–4 mm) is resected. The epidural scar tissue is dissected cranially, caudally and somewhat medially to make the plane wider and then the working sheath is moved forward and rotated to further separate the scar tissue and detect disc fragments. When sufficient laminar window already had been created at previous operation and scar adhesion was not serious, further resection of bone might not be needed. In this study, the interlaminar window was not extended in 4 among 24 patients.
Interlaminar approach has two routes: (I) via the shoulder, with working sheath inserted at the shoulder lateral to the nerve root or (II) via the axilla, with the working sheath placed at the axilla between the nerve root and dural sac. The shoulder approach is more frequently selected for shoulder and ventral type disc herniation. However, for large axillary type disc herniations, forceful retraction of the nerve root during manipulation of the disc fragments may lead to complications, such as persistent numbness or transient muscular weakness. The axilla approach is frequently chosen for the axillary type disc herniation. After removing part of herniated disc material at axilla, the tension of nerve root is reduced and the protruded disc tissue will be observed from the shoulder and the axilla, and the working sheath and endoscope can then be safely shifted to the shoulder for removing residual disc fragment. Methylene blue can be used to dye the nucleus pulposus (NP) and the displaced disc fragment, and the stained blue disc material facilitates easier visualization (22). The re-herniated fragments usually are gathered around the annular defect and not adherent to the neural tissue, and they could be removed without causing dural tear or neural damage (16). However, adhesion on the ventral side of dura sac and nerve root really occurred in our two dural tear cases, and the dura tear was not detected until the late stage of procedure when working sheath was derotated and withdrawn from the neural structure. The dural tear was small (less than 2 mm in diameter) and repair was not performed in these 2 cases. Both patients experienced transient radiculopathy without delayed CSF leakage noted and recovered uneventfully. The incidence of dural tear in revision open spine surgery is 8–18% compared with an incidence of 3.1% in primary surgery (24), and dural injury mostly occurs during dissection of the scar tissue from the neural tissue (25). With endoscopic interlaminar revision discectomy, a dural tear has occurred in 2 of 41 patients (16), in 1 of 50 patients (15) and in 0 of 10 patients (22). During endoscopic discectomy, the scar tissue may be partially dissected from the neural tissue because of continuous irrigation of water and better visualization of anatomical structures, and this may explain the lower rate of dural injury by using the endoscopic method. Increased scarring of the epidural space may be problematic, which may become clinically symptomatic, and lead to tethering of the cauda equina by postoperative connection between the epidural space and paravertebral musculature (15,26). In most cases scarring may not cause symptoms, but a small amount of herniated disc material may lead to nerve compression when the space for neural structure is severely restricted by the adhesive scar tissue. The symptoms usually improve after removal the herniated disc. Even if the scar tissue has been removed clearly, the neural structure still at the risk of adhesion after repeated surgery. Therefore, comprehensive dissection of scar tissue from the neural tissue should not be overemphasized; on the other hand, preservation of the adherent scar tissue may protect the neural tissue from injury during discectomy.
In revision spinal surgery, an extended muscle dissection and laminectomy with further facetectomy could cause segmental instability and postoperative low back pain (27). Compared to conventional procedures, tissue traumatization can be reduced to a minimum by endoscopic methods, in most cases less than 4 mm osseous margin is required to remove, and spinal stability can be preserved. There are several concerns regarding revision discectomy without fusion. One concern is that there is some inherent instability that led to the RLDH. Another concern is that revision discectomy may result in more instability when additional lamina and facet have been resected. Age is another factor to determine the success of endoscopic revision discectomy. The older ages of the patients may be associated with poor outcome (22). Ahn reported that patients younger than 40 years had 100 percent excellent or good outcomes but only 71 percent of the patients older than 40 years had such outcomes (20). The clinical outcome in the present series showed the similar results. Patients younger than 50 years had 100 percent excellent outcomes at the one year and two years follow-up. In the older patients, degeneration of the disc may occur not only in the operated level but also in the other lumbar discs, and are more likely to cause postoperative back pain or segmental instability. However, each patient’s condition and demanding varied considerably and it is difficult to make a decision just based on the age only. Additional fusion procedure may be considered in patients with instability, deformity, or associated axial low back pain, especially in the older age.
The limitation of this article is that the number of patients is small with a short-term follow-up period. The indications and effectiveness of FEID could be determined through a long-term follow-up and comparative studies with a large number of patients.
Conclusions
Recurrent lumbar herniated disc can be removed by using of the full-endoscopic technique. For the safety, identification of the bony margin and dissection of scar tissue from the medial facet joint is the key step during procedure to decrease the risk of neural injury. Full-endoscopic interlaminar approach is a safe and effective alternative to open revision discectomy.
Acknowledgments
The authors greatly appreciate the contribution of Miss Ya-Wen Wu for the assistance in clinical data collection.
Funding: None.
Footnote
Provenance and Peer Review: This article was commissioned by the Guest Editors (Hisashi Koga and Alf Giese) for the series “Full-endoscopic Spine Surgery” published in Journal of Spine Surgery. The article was sent for external peer review organized by the Guest Editors and the editorial office.
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/jss-19-370). The series “Full-endoscopic Spine Surgery” was commissioned by the editorial office without any funding or sponsorship. The authors have no other conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The trial was conducted in accordance with the Declaration of Helsinki and the Harmonized Tripartite Guideline for Good Clinical Practice from the International Conference on Harmonization. This study (IRB number: B10901026) was reviewed by the Institutional Review Board (IRB) of Buddhist Dalin Tzu-Chi General Hospital Foundation and written informed consent was obtained from all patients.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Crock HV. Observations on the management of failed spinal operations. J Bone Joint Surg Br 1976;58:193-9. [Crossref] [PubMed]
- Ebeling U, Kalbarcyk H, Reulen HJ. Microsurgical reoperation following lumbar disc surgery. Timing, surgical findings, and outcome in 92 patients. J Neurosurg 1989;70:397-404. [Crossref] [PubMed]
- Law JD, Lehman RA, Kirsch WM. Reoperation after lumbar intervertebral disc surgery. J Neurosurg 1978;48:259-63. [Crossref] [PubMed]
- Abdu RW, Abdu WA, Pearson AM, et al. Reoperation for recurrent intervertebral disc herniation in the Spine Patient Outcomes Research Trial: analysis of rate, risk factors, and outcome. Spine (Phila Pa 1976) 2017;42:1106-14. [Crossref] [PubMed]
- Kim KT, Park SW, Kim YB. Disc height and segmental motion as risk factors for recurrent lumbar disc herniation. Spine (Phila Pa 1976) 2009;34:2674-8. [Crossref] [PubMed]
- Barth M, Diepers M, Weiss C, et al. Two-year outcome after lumbar microdiscectomy versus microscopic sequestrectomy: part 2: radiographic evaluation and correlation with clinical outcome. Spine (Phila Pa 1976) 2008;33:273-9. [Crossref] [PubMed]
- Barth M, Weiss C, Thomé C. Two-year outcome after lumbar microdiscectomy versus microscopic sequestrectomy: part 1: evaluation of clinical outcome. Spine (Phila Pa 1976) 2008;33:265-72. [Crossref] [PubMed]
- Thomé C, Barth M, Scharf J, et al. Outcome after lumbar sequestrectomy compared with microdiscectomy: a prospective randomized study. J Neurosurg Spine 2005;2:271-8. [Crossref] [PubMed]
- Guan J, Ravindra VM, Schmidt MH, et al. Comparing clinical outcomes of repeat discectomy versus fusion for recurrent disc herniation utilizing the N2QOD. J Neurosurg Spine 2017;26:39-44. [Crossref] [PubMed]
- Wang JC, Dailey AT, Mummaneni PV, et al. Guideline update for the performance of fusion procedures for degenerative disease of the lumbar spine. Part 8: lumbar fusion for disc herniation and radiculopathy. J Neurosurg Spine 2014;21:48-53. [Crossref] [PubMed]
- Connolly ES. Surgery for recurrent lumbar disc herniation. Clin Neurosurg 1992;39:211-6. [PubMed]
- Swartz KR, Trost GR. Recurrent lumbar disc herniation. Neurosurg Focus 2003;15:E10-E13. [Crossref] [PubMed]
- Dai LY, Zhou Q, Yao WF, et al. Recurrent lumbar disc herniation after discectomy: outcome of repeat discectomy. Surg Neurol 2005;64:226-31; discussion 231. [Crossref] [PubMed]
- Hoogland T, van den Brekel-Dijkstra K, Schubert M, et al. Endoscopic transforaminal discectomy for recurrent lumbar disc herniation: a prospective, cohort evaluation of 262 consecutive cases. Spine 2008;33:973-8. [Crossref] [PubMed]
- Ruetten S, Komp M, Merk H, et al. Recurrent lumbar disc herniation after conventional discectomy: a prospective, randomized study comparing full-endoscopic interlaminar and transforaminal versus microsurgical revision. J Spinal Disord Tech 2009;22:122-9. [Crossref] [PubMed]
- Shin KH, Chang HG, Rhee NK, et al. Revisional percutaneous full endoscopic disc surgery for recurrent herniation of previous open lumbar discectomy. Asian Spine J 2011;5:1-9. [Crossref] [PubMed]
- Ruetten S, Komp M, Merk H, et al. Full-endoscopic interlaminar and transforaminal lumbar discectomy versus conventional microsurgical technique: a prospective, randomized, controlled study. Spine (Phila Pa 1976) 2008;33:931-9. [Crossref] [PubMed]
- Choi G, Lee SH, Lokhande P, et al. Percutaneous endoscopic approach for highly migrated intracanal disc herniations by foraminoplastic technique using rigid working channel endoscope. Spine (Phila Pa 1976) 2008;33:E508-15. [Crossref] [PubMed]
- Yeung AT, Yeung CA. Minimally invasive techniques for the management of lumbar disc herniation. Orthop Clin North Am 2007;38:363-72. [Crossref] [PubMed]
- Ahn Y, Lee SH, Park WM, et al. Percutaneous endoscopic lumbar discectomy for recurrent disc herniation: surgical technique, outcome, and prognostic factors of 43 consecutive cases. Spine (Phila Pa 1976) 2004;29:E326-E332. [Crossref] [PubMed]
- Kim CH, Chung CK, Sohn S, et al. The surgical outcome and the surgical strategy of percutaneous endoscopic discectomy for recurrent disk herniation. J Spinal Disord Tech 2014;27:415-22. [Crossref] [PubMed]
- Kim CH, Chung CK, Jahng TA, et al. Surgical outcome of percutaneous endoscopic interlaminar lumbar diskectomy for recurrent disk herniation after open diskectomy. J Spinal Disord Tech 2012;25:E125-33. [Crossref] [PubMed]
- Herron L. Recurrent lumbar disc herniation: results of repeat laminectomy and discectomy. J Spinal Disord 1994;7:161-6. [Crossref] [PubMed]
- Cammisa FP Jr, Girardi FP, Sangani PK, et al. Incidental durotomy in spine surgery. Spine (Phila Pa 1976) 2000;25:2663-7. [Crossref] [PubMed]
- Martin BI, Mirza SK, Comstock BA, et al. Reoperation rates following lumbar spine surgery and the influence of spinal fusion procedures. Spine (Phila Pa 1976) 2007;32:382-7. [Crossref] [PubMed]
- Katz JN, Lipson SJ, Larson MG, et al. The outcome of decompressive laminectomy for degenerative lumbar stenosis. J Bone Joint Surg Am 1991;73:809-16. [Crossref] [PubMed]
- Ozgen S, Naderi S, Ozek MM, et al. Findings and outcome of revision lumbar disc surgery. J Spinal Disord 1999;12:287-92. [Crossref] [PubMed]


