
Full-endoscopic debridement and drainage treating spine infection and psoas muscle abscess
Introduction
Spine infections are relatively rare, account for 2% to 4% of bone infections but the incidence is increasing due to increases in life-expectancy, risk factors, and immunocompromised patients in recent years (1-3).
Most typical clinical manifestations of spine infection is back or flank pain with or without fever episode (1,4). Neurological deficit may be presented in patients with epidural abscess or advanced spondylodiscitis with spinal canal compromise. White blood cell (WBC) may not be elevated but C-reactive protein (CRP) and erythrocyte sedimentation rate (ESR) are usually high (1,4,5). Plain film may be normal in early stage until endplate destruction or vertebral body involvement appeared. Magnetic resonance imaging (MRI) is image study of choice with approximate high sensitivity and specificity (96% and 93%) (6-9). Delayed diagnosis and missed-diagnosis is not uncommon in our daily practice. Treatment goal including obtaining causative microorganism promptly, appropriate antibiotics treatment, providing good spine stability and reduce complications as possible (10,11).
There are various treatment strategy including conservative treatment, CT-guided drainage and surgery. Conservative treatment includes bed rest, pain control and appropriate antibiotics and bracing. Average period of conservative treatment takes usually 3 months but patient sustain long period of back pain and disability even when infection has been controlled. Besides, prolong bed rest may decrease heart-lung function and ambulation capability for elderly patients. Moreover, failed conservative treatment is not uncommon in certain groups such as the elderly and immunocompromised patients. Surgical intervention is indicated when conservative treatment fails or neurological deficit appears (4,12,13).
Surgical approaches for spondylodiscitis are variable including anterior, posterior and combined approaches but optimal ones are still controversial (14-16). Traditionally, anterior open debridement, fusion followed by posterior instrumentation “Hong Kong operation” is gold standard to treat thoracolumbar spondylodiscitis (15,17,18).
In recent years, full-endoscopic debridement and drainage (FEDD) is gaining popularity because of following advantages: higher culture rate, lower anesthesia risk and minimally invasiveness (19-23).
Major advantage of FEDD over traditional anterior open surgery is that FEDD can be done under local anesthesia. Especially for elderly patients or those with multiple medical comorbidities who possess high risk undergoing general anesthesia (11,24). Besides, for patients whose infection focus located at thoracolumbar junction, traditional anterior open transpleural approach is relative major surgery and some major complications may encountered. Furthermore, many young surgeons may not familiar with this kind of surgical technique. Generally, FEDD possess less surgical wound pain, quicker patient recovery and less approach-related complications.
Since 2006, more than 150 patients with spine infection underwent surgical treatment with open anterior-posterior surgery by author and his colleagues at Taoyuan General Hospital. Since early 2015, we have shifted our surgical procedure to FEDD as first choice. Here we report our surgical technique of FEDD and preliminary clinical outcome in 34 consecutive patients.
Methods
In our institute, “Early intervention and minimally invasive surgery” is our primary strategy to dealing with spine infection. “Early intervention” means that as long as we diagnosed patients as spine infection, FEDD was done as soon as possible. We think that early “surgical” intervention with “least-invasive” endoscopic procedure could have following advantages: obtaining bacterial culture as soon as possible, early pain control, less bone-cartilage destruction and possible less hospital stay. Compared to traditional open surgery, patients and their family have higher acceptance undergoing FEDD because of its less-invasiveness and less approach-related complications. Some patients decline any operation. In these cases conservative treatment with appropriate IV antibiotics treatment is initiated.
Classification and choice of surgical approach by endoscopic surgery
Based upon the location, spine infection could be classified into two major categories including anterior or posterior pathology. Anterior groups include discitis, spondylodiscitis, psoas abscess and “anterior” epidural abscess while posterior group possess “posterior” epidural abscess and paraspinal abscess. In some circumstances, patients may sustain both pathologies (Figure 1).
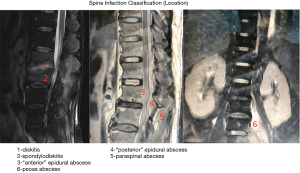
As we know, full-endoscopic transforaminal approach is originally designed to assess disc pathology. For anterior pathology group, transforaminal discectomy and drainage is an optimal approach targeting anterior column directly without destructing posterior structure. On the other hand, posterior (interlaminar) approach is suitable for “posterior” epidural abscess or paraspinal abscess.
In cases with both pathologies, we could utilize both approaches simultaneously. We suggest endoscopic spine surgeons should be familiar with both surgical approaches.
Surgical technique (author’s preference)
Transforaminal approach
There are two types of transforaminal approach including “inside-out” or “outside-in” approach. The inside-out technique, which was refined and popularized by Dr. Anthony Yeung, is that working sleeve was inserted directly into the disc space. This approach provides direct access to the inside structure of disc thus discectomy or debridement could be done. After withdrawing of working sleeve and scope into epidural space, the structure “outside” the disc such as dura or nerve root, could be addressed.
On the other hand, “outside-in” approach combined with foraminoplasty is different. Other than direct into disc space, the docking point of sleeve is on the superior articular process (SAP) of “outside-in” approach. Foraminoplasty is utilized by rasper or endoscopic drill to enlarge foramen space after removing part of ventral facet and surrounding ligament. Discectomy or debridement of disc space (inside job) is performed after foraminoplasty.
Both of these two approaches could be used but “inside-out” may be more convenient and easier in treating spondylodiscitis.
Patient was put in prone position with radiolucent operation table under local or intubated anesthesia. When the needle is introduced into involved disc space under fluoroscopic guidance, a negative-pressure syringe was adopted to see if pus was drained. The punctual needle should be advanced gradually and C-arm fluoroscopy should be checked more frequently to avoid too-deep puncture since the involved disc became softer comparing to normal disc.
When pus was drained, culture could be collected in this timing. Sometimes, the pus is too sticky to be drained, specimen could be collected after endoscope is inserted. The disc materials and necrotic tissue was debrided thoroughly by endoscopic forceps and flexible rongeurs and sent for pathology examination.
Dura and nerve roots could be checked and probed to ensure adequate neuro-decompression. After massive normal saline irrigation with or without beta-iodine solution, a 1/8 inch or 1/4 inch Hemovac drain (1/4 inch is preferred) was placed into disc space. The depth of the Hemovac drain tip could be monitored under C-arm fluoroscopy.
If the involved disc space is narrowed (disc height less than 4 mm) or concomitant foraminal stenosis is accompanied, “outside-in” approach with foraminoplasty could be adopted (Figure 2).
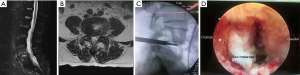
This approach could utilized stating from thoracic to lower lumbar level although thoracic spondylodiscitis is less frequent compared to lumbar spine.
Trocar-rotating method for debridement of psoas abscess
If the patient sustained concomitant psoas abscess, we could debride psoas abscess simultaneously using “Trocar-Rotating Method”, which was innovated by author and his colleague (Dr. HK Wu).
After finishing procedure within disc space, the endoscope was retracted just outside disc space where psoas muscle situated. The outer trocar “rotating” ventrally and laterally gradually with the endoscope in situ to seek for the psoas abscess. The tip of trocar should be monitored by C-arm fluoroscopy and do not advanced beyond anterior -vertebral -body -line in order not to injure great vessels. We could debride necrotic tissue and do irrigation within psoas muscle under endoscope. Movement of endoscope tip should be gentle and smooth not to injure surrounding structure such as ureter or lumbar plexus. After irrigation, a 1/4 inch Hemovac drain was placed (Figure 3).
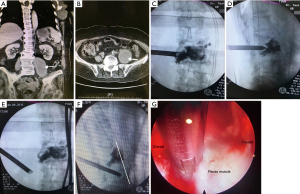
Interlaminar approach
For patients with posterior epidural abscess or paraspinal abscess, posterior interlaminar approach is an optimal approach. My preferred method is “two portal” approach. In cases of posterior epidural abscess, first portal was created and endoscopic burr was used to remove lower part of lamina. An endoscopic scissors was used to create an opening on ligamentum flavum. The opening was enlarged by scissors and kerrison punch for flavectomy. After flavectomy, pus will be encountered at epidural space and specimen could be collected. Another portal was created about 3 centimeters away from first portal and one low-pressure, flexible suction tube could be introduced to facilitate pus drainage. This technique is very useful in patients with multiple-level involvement. After drainage, a hemovac drain was placed.
Instrumentation
In uncomplicated cases with stable spine segment, FEDD without instrumentation is operation of choice. However, in complicated cases with segmental instability, instrumentation was suggested to provide better stability. Screws could be used either by traditional open method or MIS technique depends upon patient’s economy status. Decisions on posterior open surgery vs. MIS technique because of the health insurance system in Taiwan depend upon the patient’s economy status. Percutaneous pedicle screws system is not covered by health insurances in our country and patient are required to pay (about 3,000 USD for 4 screws and 2 rods). Instrumentation could be performed simultaneously with FEDD procedure using the same prone position instead of changing from decubitus position when using anterior-lateral approach (Figure 4). This is time-saving and convenient for operation team.
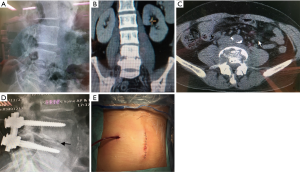
Postoperative care
After surgery, appropriate intravenous antibiotics administration was continued. Laboratory examination such as WBC, ESR and CRP were followed periodically. Patients were put on bracing or corset and ambulation is encouraged. The total amount of fluid from hemovac drain was recorded every day. Hemovac drains were removed if the drained volume was less than 10 to 15 mL per day. Patients could be discharged if back pain and laboratory data improved. Continuous oral antibiotics intake and regular out-patient follow-up was arranged.
Clinical outcomes were assessed by physical examination, laboratory findings periodically, and imaging studies. Infection was controlled with the definition of afebrile and normal laboratory findings within one year follow-up.
Results
Since June 2016 to June 2018, 34 patients including 22 males and 12 females with average aged 62.3 years (21 to 82 years old) sustained spine infection were treated by FEDD. Twenty-eight patients belong to primary spondylodiscitis and 15 patients among them sustained concomitant psoas abscess. Five patients was post-operative infection and one patient was mixed infection including lumbar spondylodiscitis, psoas abscess and multi-level posterior epidural abscess extending to cervical-thoracic spine (Table 1).
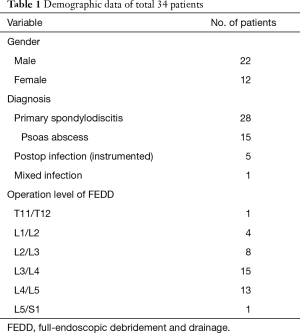
Full table
Operation level ranged from TL junction (T11/T12) to lumbosacral junction (L5/S1) while L3/4 and L4/5 were most frequent level (28/34=82%).
Twenty-six patients underwent FEDD surgery only while other 8 patients received additional posterior instrumentation. Regarding surgical approach, 32 patients underwent transforaminal approach to debride disc space and anterior vertebral bodies while 15 patients among then received “trocar-rotating method” to address psoas abscess. One patient underwent interlaminar approach for his L5/S1 spondylodiscitis. Simultaneous transforaminal and interlaminar approach was utilized in one particular patient because of mixed spondylodiscitis and posterior epidural abscess infection. Average operation time (endoscope) was 50 minutes (35 to 75 min) for each level.
There was no major intra-operative complication such as neurovascular injury or dura tear expect two patients sustained transient paresthesia and one instrument broken. Superficial wound infection was encountered in 2 patients and healed after local debridement.
Positive culture was obtained in 27 patients (27/34=79%) while gram-positive cocci (Staphyloccocci and Streptoccoi) are the most frequent pathogen (67%). Other microorganism includes E. coli, pseudomonas, Samonella, tuberculosis and non-tuberculosis mycobacteria (NTM).
Among 34 enrolled patients with minimum 12 months follow-up, infection was controlled in 28 patients (28/34=82%). Among these 6 patients with infection recurrence, two patients underwent repeat FEDD surgery and other 4 underwent open revision surgeries. Those who received repeat FEDD revealed relative stable spine segment without remarkable endplate destruction. Repeated FEDD surgery was performed via the previous approach and there were no approach-related complications. It is noticeable that these 6 unsuccessful cases, three of them were HIV positive carrier using heroin and other 3 were post-op infection with implants retention.
Overall, the 28 patients who successfully treated with FEDD were satisfied with the clinical results. Back pain or flank pain improved a lot usually within one week postoperative period. Patients who underwent FEDD surgery only without instrumentation experienced little surgical wound pain and none of them need morphine consumption. At the final follow-up period, 3 patients underwent subsequent revision posterior fusion surgery due to persisted back pain due to unstable spine segment. Two patients expired due to heart failure and terminal cancer illness at postoperative 6 weeks and 14 weeks. No recurrent infection was found among these patients during at least 12 months.
Discussion
Spine infection involves variously, from disc space to spinal canal and even spreading systemically. High morbidity or mortality rate may potentially exit if not treated well. We think early intervention with endoscopic surgery could obtain early pain control, less approach-related complications and earlier recovery.
In our series, 34 patients receiving FEDD surgery yielded high culture rate (79%) and good infection control rate (82%). Low intraoperative complications and approach-related morbidity was also evident. Surgical wound pain was reduced remarkably compared to open surgery and no morphine consumptions postoperatively because of small incision wound (8mm) by endoscopic surgery.
Our result is similar with one meta-analysis and systemic review by Mao et al. (25). The pooled event rate was 82% (95% CI: 75–88%) for positive bacteria culture, 81% (95% CI: 73–87%) for pain control satisfaction, and 21% (95% CI: 15–29%) for reoperation.
Numerous literature citations showed FEDD has higher biopsy success rate (20,26,27). Furthermore, Yang et al. (28) has proven that FEDD surgery yields higher culture rates than CT-guided biopsy (90% versus 47%). Similarly, our patients receiving FEDD surgery revealed high culture rate (79%). Compared to CT-guided biopsy using smaller puncture needle, endoscope has larger-diameter working portal (at least 3.7 mm). Other than blind procedure, FEDD surgery could collect more specimens including disc materials and necrotic tissue by endoscopic forceps and ronguers under direct vision. Besides, some microorganism could also be washed out by normal saline. We think that is why FEDD surgery is more successful in identifying the infectious agent than CT-guided biopsy because of above reasons.
However, it is noticeable that 6 failure patients in our series were more complicated initially. Three patients were HIV carrier and heroin users belonging to immunocompromised group. The other 3 were post-operative infection with previous instrumentation in situ. FEDD surgery in these special groups of patients had higher failure rate in our study. In Yang’s study, he report 65% success rate using FEDD treating instrumented lumbar spine infection in 20 patients (29). Infection -control rate of instrumented patients was lower than that of primary spondylodiscitis (65% versus 86%) in Yang’s other report (19). When dealing with these patients, we should inform patients and family about possible higher failure rate.
Two patients with infection recurrence underwent repeated FEDD surgery because of preserve spine stability. Secondary endoscopic surgery was performed via previous small operation scar and operation time was similar compared to first FEDD. During operation, minimal soft tissue fibrosis and scar was encountered. This minimal invasiveness of revision surgery is another advantage of FEDD. On the other hand, revision surgery for previous open anterior approach may be associated with higher approach-related complications.
We endoscopic surgeons should be familiar with both transforaminal and interlaminar approach since spine infection could be located in various location. As for psoas abscess debridement, “trocar-rotating technique” is a safe and effective method. For patients sustained more advanced infection with bone destruction, instrumentation should be added for better spinal stability. Different from traditional two -staged operation, we could do posterior instrumentation simultaneously with FEDD surgery using the same prone position. For those with long anterior column defect, however, anterior debridement and fusion is still necessary. We spine surgeons could not omit this important surgical technique.
Not only for patient-safety, we also concerned about safety of surgery team members. Some young patients sustained spine infection because of being intravenous drug users (IDU) with or without HIV infection. In this situation, surgeons and colleagues are exposed to high risk of contamination during surgery. Operation with endoscopic surgery, this kind of risk is less than open surgery.
Conclusions
Finally, FEDD is a safe and effective procedure treating various kinds of spine infection especially at lumbar spine area. We hope that, FEDD surgery will be the first-line surgery method to treat lumbar spine infection globally in the near future. More studies in large controlled trials on comparing the efficacy of conservative therapy, FEDD, and open surgical intervention are necessary.
Acknowledgments
Funding: None.
Footnote
Provenance and Peer Review: This article was commissioned by the Guest Editors (Hisashi Koga and Alf Giese) for the series “Full-endoscopic Spine Surgery” published in Journal of Spine Surgery. The article was sent for external peer review organized by the Guest Editors and the editorial office.
Conflicts of Interest: The author has completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/jss.2020.01.04). The series “Full-endoscopic Spine Surgery” was commissioned by the editorial office without any funding or sponsorship. The authors have no other conflicts of interest to declare.
Ethical Statement: The author is accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Hadjipavlou AG, Mader JT, Necessary JT, et al. Hematogenous pyogenic spinal infections and their surgical management. Spine (Phila Pa 1976) 2000;25:1668-79. [Crossref] [PubMed]
- Babic M, Simpfendorfer CS. Infections of the Spine. Infect Dis Clin North Am 2017;31:279-97. [Crossref] [PubMed]
- Petkova AS, Zhelyazkov CB, Kitov BD. Spontaneous Spondylodiscitis - Epidemiology, Clinical Features, Diagnosis and Treatment. Folia Med (Plovdiv) 2017;59:254-60. [Crossref] [PubMed]
- Rath SA, Neff U, Schneider O, et al. Neurosurgical management of thoracic and lumbar vertebral osteomyelitis and discitis in adults: a review of 43 consecutive surgically treated patients. Neurosurgery 1996;38:926-33. [Crossref] [PubMed]
- Przybylski GJ, Sharan AD. Single-stage autogenous bone grafting and internal fixation in the surgical management of pyogenic discitis and vertebral osteomyelitis. J Neurosurg 2001;94:1-7. [PubMed]
- Boden SD, Davis DO, Dina TS, et al. Postoperative diskitis: distinguishing early MR imaging findings from normal postoperative disk space changes. Radiology 1992;184:765-71. [Crossref] [PubMed]
- Dunbar JA, Sandoe JA, Rao AS, et al. The MRI appearances of early vertebral osteomyelitis and discitis. Clin Radiol 2010;65:974-81. [Crossref] [PubMed]
- Yang H, Wang R, Luo T, et al. MRI manifestations and differentiated diagnosis of postoperative spinal complications. J Huazhong Univ Sci Technolog Med Sci 2009;29:522-6. [Crossref] [PubMed]
- Van Goethem JW, Parizel PM, van den Hauwe L, et al. The value of MRI in the diagnosis of postoperative spondylodiscitis. Neuroradiology 2000;42:580-5. [Crossref] [PubMed]
- Karadimas EJ, Bunger C, Lindblad BE, et al. Spondylodiscitis. A retrospective study of 163 patients. Acta Orthop 2008;79:650-9. [Crossref] [PubMed]
- Chen HC, Huang TL, Chen YJ, et al. A Minimally Invasive Endoscopic Surgery for Infectious Spondylodiscitis of the Thoracic and Upper Lumbar Spine in Immunocompromised Patients. Biomed Res Int 2015;2015:780451.
- Friedman JA, Maher CO, Quast LM, et al. Spontaneous disc space infections in adults. Surg Neurol 2002;57:81-6. [Crossref] [PubMed]
- Klockner C, Valencia R. Sagittal alignment after anterior debridement and fusion with or without additional posterior instrumentation in the treatment of pyogenic and tuberculous spondylodiscitis. Spine (Phila Pa 1976) 2003;28:1036-42. [Crossref] [PubMed]
- Zarghooni K, Rollinghoff M, Sobottke R, et al. Treatment of spondylodiscitis. Int Orthop 2012;36:405-11. [Crossref] [PubMed]
- Guerado E, Cervan AM. Surgical treatment of spondylodiscitis. An update. Int Orthop 2012;36:413-20. [Crossref] [PubMed]
- Duarte RM, Vaccaro AR. Spinal infection: state of the art and management algorithm. Eur Spine J 2013;22:2787-99. [Crossref] [PubMed]
- Ito M, Abumi K, Kotani Y, et al. Clinical outcome of posterolateral endoscopic surgery for pyogenic spondylodiscitis: results of 15 patients with serious comorbid conditions. Spine (Phila Pa 1976) 2007;32:200-6. [Crossref] [PubMed]
- Nolla JM, Ariza J, Gomez-Vaquero C, et al. Spontaneous pyogenic vertebral osteomyelitis in nondrug users. Semin Arthritis Rheum 2002;31:271-8. [Crossref] [PubMed]
- Yang SC, Chen WJ, Chen HS, et al. Extended indications of percutaneous endoscopic lavage and drainage for the treatment of lumbar infectious spondylitis. Eur Spine J 2014;23:846-53. [Crossref] [PubMed]
- Fu TS, Chen LH, Chen WJ. Minimally invasive percutaneous endoscopic discectomy and drainage for infectious spondylodiscitis. Biomed J 2013;36:168-74. [Crossref] [PubMed]
- Yu WY, Siu C, Wing PC, et al. Percutaneous suction aspiration for osteomyelitis. Report of two cases. Spine (Phila Pa 1976) 1991;16:198-202. [PubMed]
- Gebhard JS, Brugman JL. Percutaneous discectomy for the treatment of bacterial discitis. Spine (Phila Pa 1976) 1994;19:855-7. [Crossref] [PubMed]
- Haaker RG, Senkal M, Kielich T, et al. Percutaneous lumbar discectomy in the treatment of lumbar discitis. Eur Spine J 1997;6:98-101. [Crossref] [PubMed]
- Fu TS, Yang SC, Tsai TT, et al. Percutaneous endoscopic debridement and drainage in immunocompromised patients with complicated infectious spondylitis. Minim Invasive Ther Allied Technol 2010;19:42-7. [Crossref] [PubMed]
- Mao Y, Li Y, Cui X. Percutaneous Endoscopic Debridement and Drainage for Spinal Infection: Systemic Review and Meta-Analysis. Pain Physician 2019;22:323-30. [PubMed]
- Li J, Yan D, Duan L, et al. Percutaneous discectomy and drainage for postoperative intervertebral discitis. Arch Orthop Trauma Surg 2011;131:173-8. [Crossref] [PubMed]
- Iwata A, Ito M, Abumi K, et al. Fungal spinal infection treated with percutaneous posterolateral endoscopic surgery. J Neurol Surg A Cent Eur Neurosurg 2014;75:170-6. [Crossref] [PubMed]
- Yang SC, Fu TS, Chen LH, et al. Identifying pathogens of spondylodiscitis: percutaneous endoscopy or CT-guided biopsy. Clin Orthop Relat Res 2008;466:3086-92. [Crossref] [PubMed]
- Yang SC, Chiu YC, Chen HS, et al. Percutaneous endoscopic debridement and drainage for the treatment of instrumented lumbar spine infection. J Orthop Surg (Hong Kong) 2019;27:2309499019863356. [Crossref] [PubMed]


