
Prophylactic muscle flap reconstruction after complex spine surgery for degenerative disease: case series and institutional protocol
Introduction
With an aging population, the number of patients with adult spinal deformities (ASDs) is expected to increase dramatically (1,2). Adult scoliosis, which can result from residual adolescent scoliosis or de novo degenerative disease, currently affects ~6% of adults over the age of 50 (3). Conservative estimates place the current total hospital discharge costs related to spinal deformity at $75.8 billion, with complications from ASD surgery costing roughly $5.4 billion (4).
As medical care, instrumentation, and surgical techniques continue to improve, there has been a subsequent rise in the number of complex spinal fusions being performed (5,6). Despite these advances, multi-level spinal fusion and surgery for deformity correction is associated with significant risk. A recent meta-analysis reported an overall complication rate of 41.2% within 30 days after ASD surgery (7). Specifically, wound complications and surgical site infection (SSI) after multilevel spinal fusion surgery can occur in up to 20% of patients (8,9). Following infection, tissues are scarred, attenuated and slow to heal, making secondary reconstruction challenging. As a result, patient morbidity skyrockets, as do the costs from prolonged care, secondary surgeries, and readmissions (8). The risk of wound complications in multi-level spinal fusion is further compounded by obesity, diabetes, poor nutritional status, and prolonged operative time (8-13).
In high-risk patients, musculocutaneous flap coverage is a viable option to manage complex postsurgical wounds following multi-level spinal fusion (11). Flap coverage promotes wound healing by increasing vascularity, obliterating dead space, and reducing tension on opposed skin margins (11). Use of local muscle flaps has low donor site morbidity; therefore, their use in complex spine wound closure is associated with low risk and a potential for significant gain (11). Currently, the use of local flaps has been reported in high-risk patients with a history of spinal neoplasms, radiation therapy, exposed hardware, multiple spine surgeries, or wound infections (11,14-21). To the best of our knowledge, there are no prior reports on the use of prophylactic flaps in patients undergoing posterior spinal fusion for degenerative pathology. The objective of this study was to describe the indications, procedures, and outcomes of prophylactic muscle flaps for wound coverage after instrumented posterior spinal fusion for multi-level degenerative spine disease and spinal deformity.
Methods
Research design and sample
An institutional review board (IRB) approved retrospective analysis of patients who underwent multi-level posterior spinal fusion for degenerative disease and had concurrent muscle flap coverage of their wound was done. We included patients aged between 18 and 85 years old who were operated at a single academic institution between August 2016 and February 2017 with regular follow-up for at least 6 months. Primary and revision procedures of the cervical, thoracic, and/or lumbosacral spine were included. Patients who had flap coverage following oncological resection, and for management of wound dehiscence/infection were excluded. Spinal fusions were done by orthopaedic surgery board-certified spine surgeons (SN Khan, E Yu) and flap coverage was done by a board-certified plastic surgeon (R Chandawarkar) in all cases.
Data extraction and analysis
Comprehensive review of patient’s electronic medical record (EMR) was done to record demographics such as age, gender, ethnicity, and insurance provider. Various clinical parameters such as body mass index (BMI), smoking status, American Society of Anaesthesiologists (ASA) grade, and systemic comorbidities were recorded. Finally, procedure related information such as region and levels of surgery, primary vs. revision surgery, drains used, type of flap (paraspinous, trapezius, latissimus dorsi), flap area of coverage, length of hospital stay, discharge disposition, etc. have been described. Presence of any complications such as dehiscence, infection, seroma, revision surgery and healing of flap at latest follow-up were our main outcome measures. Categorical data have been expressed as number and percentage, and quantitative data have been expressed as mean, standard deviation or range.
Flap protocol
A pre-operative referral to the plastic surgery team is placed for those patients undergoing multi-level spinal fusion and/or deformity correction for whom wound coverage and healing problems are anticipated. During this office visit, evaluation of the skin and soft tissues over the planned levels of spinal fusion is done. The indications, alternative options, rationale, benefits and risks of muscle flap coverage are discussed in detail. The risks that are discussed include but are not limited to bleeding, infection, wound breakdown, poor quality wound healing, scarring, pain, paresthesia, seroma formation, loss of the flap or tissue viability, loss of skin sensation, asymmetry, fat necrosis, contour abnormalities and revision surgery. In addition, incisions for the flaps are explained in detail using diagrams so that the patient understands the resultant scars. After the patient completely understands the procedure and all of their questions are answered, the decision for flap coverage after spine surgery is made. Depending on the extent of wound defect, the following surgical techniques are used in isolation or combination to achieve complete coverage.
Cervical spine
The trapezius, either unilaterally or bilaterally depending on requirement, is used, as the paraspinous musculature has limited mobility in this region. It is detached inferiorly, such that the dorsal scapular branch is preserved and is rotated for coverage. This is done by sharply dividing the medial portion of trapezius muscle and lifting it up from the posterior chest wall until the dorsal scapular artery is visualized. Keeping this preserved, the lower portion of the trapezius muscle is sharply divided and rotated upwards. This part of the trapezius is ready for inset and closure over to the paraspinous muscles on the opposite side. This strategy allows coverage of the hardware as well as the exposed posterior vertebral column very effectively (Figures 1,2).
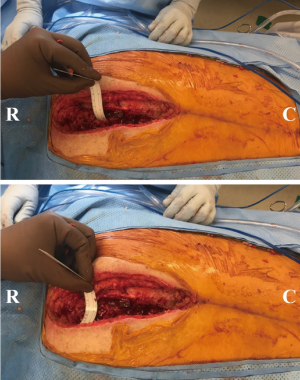
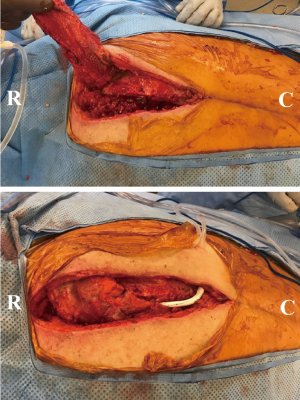
Trapezius propeller flap for cervicothoracic spine
This flap is used when coverage is required at the cervicothoracic junction and upper thoracic spine. The initial preparation involves cutting the medial most border of the trapezius and lifting it up from the posterior chest wall until the dorsal scapular artery is visualized and preserved. After this, the upper portion of the trapezius muscle (only the vertical and lower part of the horizontal fibers) is sharply divided. The vertical fibers and the upper horizontal fibers of the trapezius muscle are carefully preserved to minimize the risk of functional deficit. The dorsal scapular vessels are dissected free into the fascia to allow adequate rotation for a propeller flap. Care is taken to ensure that the vascular pedicle was not kinked or twisted. The propeller flap is then rotated 160 degrees counter-clockwise and closure over to the paraspinous muscles on the opposite side is done.
Thoracic and lumbar spine
Over the bilateral paraspinous region, dissection is performed in the fascio-cutaneous plane just above the muscle until the lateral border of the muscle is reached. Next, the lateral portion of the muscle is carefully dissected and freed so that the bilateral paraspinous muscles are lifted off minimally and advanced medially towards the midline. The lumbar vascular perforators enter from the deep and lateral aspect of the longissimus and iliocostalis, and need to be fully preserved for flap advancement. The lumbar perforators (three on left and four on the right) need to be carefully dissected circumferentially to preserve the blood supply to the flap and allow maximum excursion of the flap medially. In addition, the fascio-cutaneous flaps are secured and advanced medially with quilting sutures in two separate rows on each side to reduce tension and help adherence of the flaps to the underlying musculature and reduce the risk of seroma formation. Using eight 2-0 Maxon™ (Medtronic, Minneapolis, MN, USA) sutures (four in each row), the fascia on each side is mobilized and sutured down to the underlying muscle to obliterate the dead space. After this, the skin edges are easily approximated without any tension, followed by 2-0 V-Loc™ (Medtronic, Minneapolis, MN, USA) and staples on the skin. Two number 15 hubless drains are placed in this area and fixed (one under the skin flap and one under the muscle flaps) (Figures 3-5).
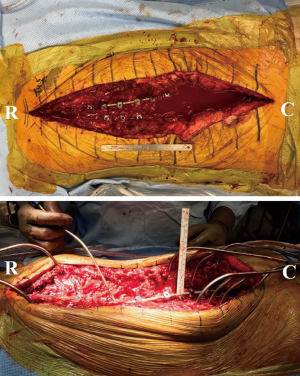
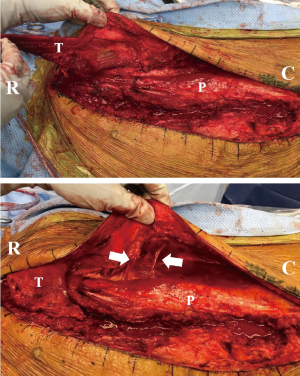
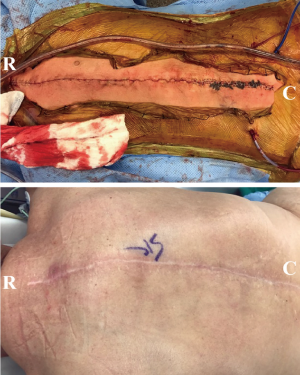
Results
A total of 26 patients with a mean age of 59.7±13.0 years were included, out of which 73.1% (n=19) were females (Table 1). The mean BMI of the patient cohort was 31.0±8.6 kg/m2. Majority of the patients had either quit smoking more than 6-month prior to surgery (n=11, 42.3%) or never smoked (n=9, 34.6%). Prevalent comorbidities in the patient cohort included hypertension (n=15, 57.7%), hyperlipidemia (n=10, 38.5%) and diabetes (n=5, 19.2%). Two patients had a history of malignancy but without any metastases to the spine. The clinical profile of the patient cohort has been summarized in Table 1.

Full table
A majority of patients (n=18, 69.2%) were undergoing a revision spinal fusion. Out of these, 4 (22.2%) patients had a history of SSI during primary fusion. However, their SSI had been cleared with complete healing of the wound before the revision fusion was indicated. All fusions were instrumented and were performed through the open posterior approach. The mean number of levels of fusion was 10.2±4.5 with majority fusions extending from the thoracic spine to the pelvis (n=15, 57.7%) (Table 2).

Full table
Isolated paraspinous muscle flaps were used in a majority of the cases (n=16, 61.5%), and paraspinous combined with trapezius flaps were used in 9 cases (34.6%). The average area of defect that was covered with flaps was 325 cm2, with an average length of 23 cm (range, 10–36 cm). Flap drains were removed at mean 24.4±10.6 days after surgery, with a range of 5 to 47 days (Table 2).
All wounds (100%) completely healed with no complications at latest follow-up (average 9.1 months; range, 6–16 months). One patient developed a seroma for which interventional radiology (IR) assisted drainage was sufficient.
Discussion
Multi-level spinal fusions have been associated with high complication rates and roughly $290 billion in costs between 2000 and 2010 for approximately 4 million procedures (22,23). Additionally, the risk of complications and in turn the cost of care increases as the number of levels of surgery increases (23). Known risk factors for wound complications include history of infection, revision surgery, use of instrumentation, longer operative time, greater than six operated spinal levels, and surgical approach (11,22,24). Patient comorbidities such as diabetes, obesity, and chronic obstructive pulmonary disease (COPD) have also been correlated with wound complications (8-11). Conventionally, SSI and wound complications require long-term antibiotics, additional surgery, and prolonged hospital stay for management, all of which result in increased morbidity and healthcare costs (8,11). We report our experience and protocol for prophylactic muscle flap coverage after multi-level posterior spinal fusion for degenerative conditions as a means to decrease the risk of wound complications.
Current literature on the use of immediate and delayed muscle flap closure of wounds after spine surgery involve high-risk patients such as those with history of radiation therapy, exposed hardware, multiple spine surgeries and wound dehiscence/infection, mainly after spinal neoplasm resection. Studies have reported wound complication rates ranging from 12% to 41% after delayed flap reconstruction in these high-risk patients (14,16-19). Prophylactic muscle flap coverage in 52 high-risk patients reported a 25% rate of seroma formation, 15% infection rate, 10% would dehiscence rate, and 12% surgical re-intervention rate (21). A larger study with 96 patients who received concurrent spinal fusion and flap reconstruction noted post-operative wound complications in 5.9% and a reoperation rate of 3.4% (11). Finally, results from a meta-analysis show that there is a statistically significant decrease in the incidence of complications in patients who undergo prophylactic muscle flap reconstruction when compared to those who did not (13). Immediate reconstruction of surgical wounds with muscle flaps has been show to significantly reduce the number of instrument explanations and the number of unplanned reoperations as well (12).
In our case series, muscle flaps were used for coverage during the primary closure of wounds after an open posterior approach after multi-level spinal fusion for degenerative spine disease. Revision surgery such as that for extension of previous fusion, revision of hardware or for pseudo-arthrosis are common indications to perform flap closure at our institution. Going through the scar from previous surgery is complicated by poor vascularity, limited surrounding soft tissue, and higher tension on skin edges (11,13). Another indication for which we consider flap coverage is in patients with either extremes of low or high BMI. In patients with low BMI there is concern for inadequate soft tissue coverage of the hardware and fusion bed. In patients with high BMI, there is potential for large dead space and eventually infection due to subcutaneous fat thickness (7). The use of paraspinous and trapezius muscle flaps helps improve vascularity and eliminate dead space in these situations (13). Other risk factors such as poor nutritional status and diabetes are considerations for flap closure as well. We do not routinely measure nutritional parameters in patients undergoing surgery for multi-level degenerative pathology or spinal deformity but follow a universal pre-operative protocol for nutritional supplementation in all cases. This includes advice for off the shelf protein powder shakes, 1,000 units of cholecalciferol and 1,000 mg of calcium daily.
During our 7-month study period, 26 patients underwent multi-level posterior spinal fusion and simultaneous wound closure using local muscle flaps to cover defects ranging from 98 to 648 cm2 in area. All 26 patients had complete healing of their wound at their latest follow-up. Only one patient in our group developed a seroma which was drained percutaneously without sequelae. Although in a small patient series, we believe that our results highlight a remarkable potential in reducing morbidity and costs related to wound complications after open posterior multi-level spinal fusion. Results from previous studies on muscle flap coverage in high-risk patients such as those with spinal neoplasm demonstrate a significant reduction in wound complications, although they report a higher incidence than ours. Such a difference is expected given that patients with spinal neoplasm are usually sicker and have a worse surgical risk profile than patients undergoing surgery for extensive degenerative disease.
Our study is primarily limited by its retrospective nature and small sample size. Additionally, findings from a single institution may not always be uniformly generalized. There may be variability in the support and availability of plastic surgeons across institutions nationwide for such a team approach. Given the muscle flap closure protocol in place, we were unable to report results from a comparative group undergoing standard wound closure techniques. A lower incidence of wound complications can be taken as indirect evidence for overall cost savings associated with complex spine surgery; however, analysis of cost-effectiveness was beyond the scope of this study.
Conclusions
We conclude that utilizing a team approach and protocol for management of wound coverage after multi-level spinal fusion with or without deformity correction has the potential to minimize complications and achieve excellent surgical outcomes. Additionally, such a protocol has the potential to improve the overall quality and value of care in extensive spinal fusions, which are otherwise fraught with complications. Further experience with this approach from our institution and development of individual protocols nationwide will help substantiate these preliminary results.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: The study was approved by the Institutional Review Board (IRB number: 2016H0398) and informed consent was waived.
References
- Silva FE, Lenke LG. Adult degenerative scoliosis: evaluation and management. Neurosurg Focus 2010;28. [Crossref] [PubMed]
- Somani S, Capua JD, Kim JS, et al. ASA Classification as a Risk Stratification Tool in Adult Spinal Deformity Surgery: A Study of 5805 Patients. Global Spine J 2017;7:719-26. [Crossref] [PubMed]
- Youssef JA, Orndorff DO, Patty CA, et al. Current Status of Adult Spinal Deformity. Global Spine J 2013;3:51-62. [Crossref] [PubMed]
- United States Bone and Joint Initiative. The Burden of Musculoskeletal Diseases in the United States: Prevalence, Societal and Economic Costs. 3rd ed. Rosemont, IL: United States Bone and Joint Initiative, 2015. Available online: http://www.boneandjointburden.org/
- Cowan JA Jr, Dimick JB, Wainess R, et al. Changes in the Utilization of Spinal Fusion in the United States. Neurosurgery 2006;59:15-20; discussion 15-20. [Crossref] [PubMed]
- Paulus MC, Kalantar SB, Radcliff K. Cost and Value of Spinal Deformity Surgery. Spine (Phila Pa 1976) 2014;39:388-93. [Crossref] [PubMed]
- Yadla S, Maltenfort MG, Ratliff JK, et al. Adult scoliosis surgery outcomes: a systematic review. Neurosurg Focus 2010;28. [Crossref] [PubMed]
- Lee NJ, Shin JI, Kothari P, et al. Incidence, Impact, and Risk Factors for 30-Day Wound Complications Following Elective Adult Spinal Deformity Surgery. Global Spine J 2017;7:417-24. [Crossref] [PubMed]
- Pull ter Gunne AF, van Laarhoven CJ, Cohen DB. Incidence of surgical site infection following adult spinal deformity surgery: an analysis of patient risk. Eur Spine J 2010;19:982-8. [Crossref] [PubMed]
- Schoenfeld AJ, Carey PA, Cleveland AW 3rd, et al. Patient factors, comorbidities, and surgical characteristics that increase mortality and complication risk after spinal arthrodesis: a prognostic study based on 5,887 patients. Spine J 2013;13:1171-9. [Crossref] [PubMed]
- Cohen LE, Fullerton N, Mundy LR, et al. Optimizing successful outcomes in complex spine reconstruction using local muscle flaps. Plast Reconstr Surg 2016;137:295-301. [Crossref] [PubMed]
- Devulapalli C, Broyles JM, Bello R, et al. Soft-Tissue Reconstruction of Large Spinal Defects: A 12-Year Institutional Experience. Plast Reconstr Surg 2017;140:806-14. [Crossref] [PubMed]
- Chieng LO, Hubbard Z, Salgado CJ, et al. Reconstruction of open wounds as a complication of spinal surgery with flaps: a systematic review. Neurosurg Focus 2015;39. [Crossref] [PubMed]
- Wilhelmi BJ, Snyder N, Colquhoun T, et al. Bipedicle paraspinous muscle aps for spinal wound closure: An anatomic and clinical study. Plast Reconstr Surg 2000;106:1305-11. [Crossref] [PubMed]
- Mericli AF, Tarola NA, Moore JH Jr, et al. Paraspinous muscle ap reconstruction of com- plex midline back wounds: Risk factors and postreconstruction complications. Ann Plast Surg 2010;65:219-24. [Crossref] [PubMed]
- Hultman CS, Jones GE, Losken A, et al. Salvage of infected spinal hardware with paraspinous muscle flaps: anatomic considerations with clinical correlation. Ann Plast Surg 2006;57:521-8. [Crossref] [PubMed]
- Manstein ME, Manstein CH, Manstein G. Paraspinous muscle flaps. Ann Plast Surg 1998;40:458-62. [Crossref] [PubMed]
- Saint-Cyr M, Nikolis A, Moumdjian R, et al. Paraspinous muscle flaps for the treatment and prevention of cerebrospinal fluid fistulas in neurosurgery. Spine (Phila Pa 1976) 2003;28:E86-92. [Crossref] [PubMed]
- Casas LA, Lewis VL Jr. A reliable approach to the closure of large acquired midline defects of the back. Plast Reconstr Surg 1989;84:632-41. [Crossref] [PubMed]
- Dumanian GA, Ondra SL, Liu J, et al. Muscle flap salvage of spine wounds with soft tissue defects or infection. Spine (Phila Pa 1976) 2003;28:1203-11. [Crossref] [PubMed]
- Garvey PB, Rhines LD, Dong W, et al. Immediate soft-tissue reconstruction for complex defects of the spine following surgery for spinal neoplasms. Plast Reconstr Surg 2010;125:1460-6. [Crossref] [PubMed]
- Lee MJ, Konodi MA, Cizik AM, et al. Risk factors for medical complication after spine surgery: a multivariate analysis of 1,591 patients. Spine J 2012;12:197-206. [Crossref] [PubMed]
- Puvanesarajah V, Cancienne JM, Werner BC, et al. Perioperative Complications Associated with Posterolateral Spine Fusions: A Study of Elderly Medicare Beneficiaries. Spine (Phila Pa 1976) 2018;43:16-21. [Crossref] [PubMed]
- Glassman SD, Bridwell KH, Shaffrey CI, et al. Health-Related Quality of Life Scores Underestimate the Impact of Major Complications in Lumbar Degenerative Scoliosis Surgery. Spine Deform 2018;6:67-71. [Crossref] [PubMed]


