The impact of urgent intervention on the neurologic recovery in patients with thoracolumbar fractures
Introduction
The treatment of thoracolumbar (ThL) fractures remains controversial. The main questions refer to the choice of a reliable classification system (1,2), the variability in the approach (3), the decompression technique (4,5) and the timing of the intervention, particularly if spinal cord injury (SCI) is present (6) . Based on the comprehensive AO classification system (1), the new AO Thoracolumbar Injury Classification System (TLICS) evaluates the neurologic status, integrity of the posterior ligamentous complex and injury pattern, and serves as a guide for treatment decision using a scoring system (2). For distraction and translational/rotation injuries, stabilization and indirect decompression can be performed using a posterior approach. For compression-burst injuries with posterior complex ligaments being intact and without neurologic impairment, a posterior short segment fusion, including vertebral fracture, can prevent failure. However, if epidural compression and neurologic compromise are present, an anterior direct decompression must be carried out (3,7). Unlike classical open techniques, transpedicular posterior approach and minimally invasive techniques allow adequate spinal decompression while also decreasing morbidity related to the anterior approach. However, in the case of a ThL burst fracture with an incomplete neurologic deficit, none of the surgical approaches used has demonstrated advantages over others with regard to neurologic recovery (4).
It is widely accepted that the main principle in the treatment of unstable vertebral fractures is adequate stabilization to prevent pain or secondary deformity. In addition, if a SCI is present, urgent or early treatment with epidural or intradural decompression techniques has the potential to reduce intradural pressure, increasing blood supply and prevent secondary injury mechanisms dues to ischemia (8,9). Moreover, these have proven to be beneficial in reducing the length of stay (LOS) and related complications (10). Despite the above, whether there is any improvement in neurologic status remains controversial (6). Experimental studies in animals have shown an improvement in neurologic function with early neurological decompression (11), but similar studies in humans would be unethical as it would involve randomization for surgical treatment randomization. Although demonstrating improvement in the neurologic status is challenging, recent studies recommend early surgical management in less than 24 h after injury in patients with traumatic SCI, in order to improve neurologic and functional outcomes (12-14).
Taking this into account and the fact that spinal cord functional recovery is also time dependent, the aim of this study is to describe the benefits of performing urgent (≤8 h) surgery in patients with ThL fractures and with a neurologic lesion, which could contribute to the process of decision-making in the management of such fractures in the future.
Methods
Study design and study population
We carried out an observational longitudinal retrospective study.
All patients with an unstable ThL fracture and neurological impairment who underwent surgery between 2000 and 2009 at the Hospital Spine Unit were screened. Patients with thoracic and ThL spine fractures with traumatic SCI were initially included in the study. Patients with a vertebral fracture in the lumbar spine (L3–L5), a chronic lesion (Kummel disease) and not intervened, and patients with loss of follow-up, were excluded from the study.
All patients had been evaluated on admission to the hospital by the emergency department, where their severity according to the injury severity score (ISS) to provide an overall score for patients with multiple injuries was assessed (15). The American Spinal Injury Association (ASIA) Impairment Scale was used to establish neurologic status prior to surgery (16). A high dose of methylprednisolone was used according to NASCIS II protocol following the emergency department or the orthopaedic surgeon criteria (17). An X-ray study and computed tomography (CT) were performed in all patients.
The approach, decompression technique, and levels of fusion were chosen following the morphological pattern according to the AO classification. Short or long fusion was performed and indirect decompression and stabilisation by the use of pedicular screws and structural bone graft obtained from the posterosuperior iliac spine in patients with type B flexion-distraction and type C rotational injuries. In type A burst fractures, a posterior, anterior or combined approach and decompression technique were chosen according to the load-sharing classification. We performed a combined approach if compression and distraction pattern injuries were present.
Patient follow up and data collection
Patients in the study had been followed up periodically for at least 2 years after surgery to assess their radiological, functional and neurologic status. This information was collected for the study from the registry of the emergency service at the time of the accident and data obtained from medical records and the surgical register. The patient information was entered into a database and treated confidentially.
Study variables
The following variables were analysed: age; sex; cause of injury; existence of associated lesions and severity grade (ISS); type of lesion based in the AO classification system prior to the X-ray and CT assessment; the use of high doses of methylprednisolone before surgery following the N.A.S.C.I.S III protocol; location and level of injury (thoracic spine T1–T9 or ThL junction T10–L2); type of surgical procedure performed (levels of fixation and anterior, posterior or combined approach); time elapsed between injury and surgery (≤8 and >8 h from the time of injury); LOS; and pre- and postoperative neurological status according to the ASIA scale at the time of injury and 2 years after the follow-up. Patients were categorized into classes from A to E depending on their motor and sensory functions according to the same scale, and each class was graded between 1 and 5. The worst grade, grade 1, was given to class A, and the best, grade 5, was given to class E (12).
Statistical analysis
The analysis consisted of a description of qualitative and quantitative variables using frequency distributions and measures of central tendency and variability, respectively. For the statistical analysis patients were divided into two groups: those intervened within the first 8 h after the lesion had occurred and those intervened after those first 8 h. We used the Chi-square test and the exact Fisher test when necessary to compare proportions in independent groups, the Wilcoxon test for repeated measures and the Mann-Whitney U test for mean values, all at the significance level of P<0.05. We tested the ASIA score improvement at the end of follow-up using multiple regression analysis, adjusted by variables such as ISS, timing of intervention, location, approach and type of fracture. The analysis was carried out using SPSS v 19.0 (IBM SPSS Inc., Chicago, IL, USA).
Results
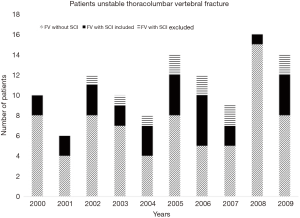
Out of a total of 111 patients with unstable ThL fractures who underwent surgery, 39 had a neurologic impairment (35.1%), of which 28 were available and included in the study (Figure 1). Eleven patients were excluded: seven were lost to follow-up due to transfer to their place of residence, discharge centre or occupational control, two not intervened, one for Kummel disease and one at lumbar level.
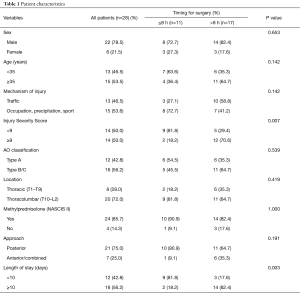
Patients in the study included 22 men (78.5%) and 6 women (21.5%), with a mean and a median of 36.8 and 35.5 years old respectively. The most common cause of the lesions was road accidents (46.4%), followed by falling (32.1%), occupational accidents (14.3%) and sports-related accidents (7.1%). The ISS mean was 11.3. According to the AO classification system, 42.8% of the patients had type A lesions (all of them being A3-burst), 17.8% had type B, and 39.3% had type C. High doses of corticoids were administered to 85.7% of the patients prior to surgery according to the NASCIS III protocol. In 28% of the patients, the location of the lesion was the thoracic spine T1–T9, whereas in the rest of the cases, the location was the ThL spine T10–L2. The posterior approach was used in 75% of the cases (21 patients) and indirect decompression was performed in all of them. Out of the 21, short fusion (less than or equal to three levels) was performed in 14 patients, using pedicle screws at the fractured vertebra in 11 of them. In the other 7 patients, we performed long fusion, with an average of 5.5 levels of fusion. In 6 (21% of the patients), we carried out an anterior direct decompression with corpectomy by thoracotomy or ThL retroperitoneal approach and interbody fusion using a structural autograft iliac crest and plate. In 1 (4%), we performed a combined approach of the two separate interventions. LOS mean was 11.5 days (Table 1). Postoperative complications were observed in three patients (5.8% of the total). Two patients with postoperative wound infections required debridement and antibiotic treatment and long fusion was performed in one patient who suffered from coronal imbalance.
Full table
An urgent surgical procedure, within the first 8 h after the injury had occurred, was carried out in 11 (39.3%) vs. 17 patients (60.7%) who underwent surgery after more than 8 h (one patient intervened at 11 h, two patients between 11–24 h, six between 25–72 h and eight after 72 h). In these two groups, no statistically significant differences were observed in age, sex, mechanism of injury, AO classification, location and approach used in both groups, while the ISS (P=0.007) and LOS (P=0.003) were statistically significant (Table 1).
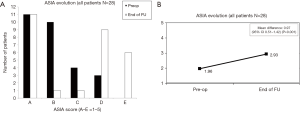
In the initial assessment prior to surgery, 39.3% of the patients had a complete lesion (ASIA A), 35.7% had an ASIA B, 14.3% had an ASIA C, and 10.7% had an ASIA D lesion. In the assessment conducted at the end of follow-up after more than 2 years, and once the discharge treatment was completed, 39.3% continued with an ASIA scale grade of A, 3.5% with an ASIA scale grade of B, 3.5% with an ASIA scale grade of C, 32.2% with an ASIA scale grade of D and 21.4% with an ASIA scale grade of E (Figure 2A). The mean ASIA scale pre-operative for all patients (ASIA A–D) was 1.96 and 2.93 at the end of follow-up with the mean difference 0.97 (95% CI, 0.51–1.42; P=0.001) (Figure 2B).
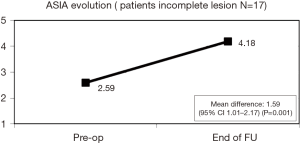
There were no changes in patients with complete lesions (ASIA A). In patients with incomplete lesions (ASIA B–D), the pre-operative ASIA scale was 2.59 and 4.18 at the end of follow-up and the mean difference was 1.59 (95% CI, 1.01–2.17; P=0.001) (Figure 3). Improved ASIA scale grades in patients with incomplete lesions (B–D) who underwent urgent surgery within less than 8 h was 1.73 and 0.47 in those intervened within more than 8 h after the injury had occurred, with a statistically significant difference 1.26 (95% CI, 0.45–2.05; P=0.007; Figure 4A,B,C).
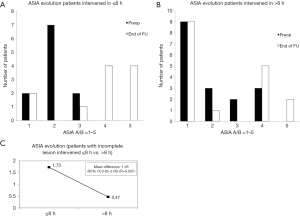
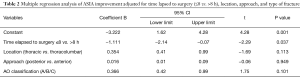
Results from the multiple regression analysis, adjusted for type and location of the fracture, approach and timing of intervention, showed that the post-operative ASIA improvement was statistically significant in patients intervened within the first 8 h compared to those intervened later (P=0.037) (Table 2).
Full table
Discussion
SCI is one of the primary causes of neurologic disability with major individual, family and economic impact. In Spain, there is an estimated incidence of 12–20 new cases per million inhabitants per year (18). During the period of our study, 111 unstable vertebral fractures were intervened, of which three to four patients presented SCI, equivalent to the above annual incidence.
There are multiple constraints associated with the management of ThL fractures with neurologic impairment, such as ethical issues excluding the possibility of conducting randomised studies and health system-related demands including patient transport, performing imaging studies and other associated lesions, as well as the prompt availability of a specialised surgical team. In addition, SCI is a relatively low frequency condition, therefore large study sample sizes are difficult to achieve. Our study represents all patients intervened through the period of 10 years, by the same surgery team and where standardised assessment was followed.
The main goal of unstable ThL fracture treatment is to achieve spine stability to prevent painful secondary deformities. If this is accompanied by neurologic impairment, then spinal cord decompression with neurologic recovery is also a goal (8). To prevent secondary injury mechanisms after an initial primary injury, medical therapies have been used. SCI in the upper thoracic region can result in a loss of sympathetic outflow resulting in neurogenic shock. Preventing secondary injury due to hypotension and hypoxia is the main goal during the resuscitation of the spinal cord injured patient. Clinical practice guidelines recommend achieving a mean arterial pressure of at least 85 mmHg, although no conclusive data support this target. Fluid resuscitation and pressers may be required (19). In our study, we used high doses of glucocorticoids in 85% of the patients during the first hours after injury according to the NASCIS II and III guidelines (17). This did not show any significant benefit in neurological recovery. In fact, they have been found to increase the risk of complications such as severe sepsis and pneumonia and thus, recent studies discourage their use (20).
Given an irreversible primary lesion and the lack of effective medical therapies, early decompression should have a neuroprotective effect (21). However, the advantages of early decompression and the best time for surgery remain controversial. Surgical decompression has the potential to reduce intradural pressure, thus increasing blood flow to the spinal cord, reducing ischemia, and preventing secondary injury mechanisms. Although these pathophysiologic changes have been shown in experimental animal studies (11), the benefits of urgent or early decompression have not been clearly demonstrated in clinical practice due to the heterogeneity of patients, type of injury and spinal location, and the difficulties associated with the performance of a prompt surgical procedure as well as with the implementation of high level of evidence studies due to ethical reasons.
Following the primary vertebro-medullary injury, the morphological pattern, approach, location of lesion and time elapsed to surgery of may influence the neurologic recovery. Through the multiple regression study, in our study the time elapsed until the intervention was shown to be a significant factor for improvement of the neurological function in patients who were intervened promptly within the first 8 h compared to those intervened after that time.
Classification systems of injury have been questioned for their reliability and reproducibility. The new AO Spine thoracolumbar spine system classification is based on the neurological lesion, morphological pattern and lesion of the posterior ligament complex evaluated by MR. In our study the MR assessment was not possible in all cases in the preoperative period, especially if urgent surgery was considered and the morphological pattern was identified by CT according to the AO classification (1). A sequential damage assessment of the posterior ligamentous complex after the MR study has been related with the fracture morphological pattern evaluated by TC study (22). The morphological pattern was not associated with neurological improvement in our analysis.
The morphology pattern of the lesion should be used to determine the best approach (3). Flexion-distraction and rotational injuries were treated by posterior approach and for the burst fractures we followed the load-sharing classification to choose between anterior, posterior or combined approaches and the use of a decompression technique. More recently, the technique of transpedicular decompression using the posterior approach has been found to allow less morbidity, thus avoiding the anterior approach. In addition to extradural decompression techniques, surgical intradural decompression by durotomy has the potential to reduce intradural pressure and therefore increases blood flow to the spinal cord, reduces ischemia and prevents secondary injury mechanisms (23). However, in our study the approach and decompression technique were not statistically associated with the neurologic improvement, in accordance with recent study which reported that there is no specific surgical approach in the case of a ThL burst fracture with an incomplete neurologic deficit having any advantage as far as neurologic recovery is concerned (4,5). Moreover, the level of injury and ASIA neurologic scale impairment can determine the functional recovery prognosis due to anatomic considerations and the blood supply. In our patients, thoracic or ThL lesions were not statistically associated with the neurologic improvement despite thoracic fractures presenting a worse prognosis than ThL and lumbar fractures (24).
Based on the current literature, there is evidence that early surgical stabilisation leads to shorter hospital stays, shorter intensive care unit stays, and lower pulmonary complications and costs, all the more evident in patients who have more severe associated injuries as measured by the ISS. In both patients with and without SCI, early surgical stabilisation has also been found to be beneficial (10). Furthermore, there is some evidence that early stabilisation does not increase complication rates compared with late surgery (25). In addition, the best timing for surgery following an SCI remains controversial. Although previous studies have not proven that early intervention does influence the neurologic improvement (26) a systematic review of clinical studies concluded that decompress surgery performed within the first 24 h resulted in superior clinical outcomes compared with decompression performed after the first 24 h (27). Fehlings et al., in another systematic review study, collected opinions from 971 surgeons and concluded that early surgery, when performed in less than 24 h, is strongly recommended in the event of neurologic injury but before 12 h in cases of incomplete SCI (28). Wilson et al., in a prospective cohort study showed a statistically significant difference in patients who received early surgery (<24 h) vs. late (14). Cengiz et al., in a prospective study of 27 patients with ThL injuries, showed that twelve patients having undergone surgery within 8 h had greater neurologic recovery in comparison with 15 patients who underwent surgery within 3 to 15 days (12).
In our study, no changes were seen in patients with complete lesions ASIA A scale at preoperative and in the follow up assessments. However, the ASIA scale was statistically associated with the neurologic improvement, so patients with incomplete lesions (ASIA scale B–D) intervened urgently in less than 8 h had greater neurologic improvement compared with those patients with more delayed surgery (1.26, P=0.007), which could determine changes in functional status. The multiple regression analysis also showed statistically significant differences in the ASIA scale with neurologic improvement at the end of follow-up when the time elapsed to surgery was within first eight (P=0.024). These results suggest, as other studies mentioned above, that early surgery should be performed as soon as possible to improve the neurological status of the patients and if possible to intervene within the first 8 h after injury.
Conclusions
Our study showed that in patients with an incomplete SCI, there was significant improvement in patients intervened early, particularly if this was performed within the first 8 h after the injury occurred. Given the devastating consequences in the functional status of these patients, there should be a common effort to achieve the earliest timing of intervention possible.
Acknowledgements
Thanks to Ana, Alejandro and Javier for their help.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: The study was approved by an Ethics Committee of the institution within which the work was undertaken and that it conforms to the provisions of the Declaration of Helsinki.
References
- Magerl F, Aebi M, Gertzbein SD, et al. A comprehensive classification of thoracic and lumbar injuries. Eur Spine J 1994;3:184-201. [Crossref] [PubMed]
- Vaccaro AR, Oner C, Kepler CK, et al. AOSpine thoracolumbar spine injury classification system: fracture description, neurological status, and key modifiers. Spine (Phila Pa 1976) 2013;38:2028-37. [Crossref] [PubMed]
- Vaccaro AR, Lim MR, Hurlbert RJ, et al. Surgical decision making for unstable thoracolumbar spine injuries: results of a consensus panel review by the Spine Trauma Study Group. J Spinal Disord Tech 2006;19:1-10. [Crossref] [PubMed]
- Oner FC, Wood KB, Smith JS, et al. Therapeutic decision making in thoracolumbar spine trauma. Spine (Phila Pa 1976) 2010;35:S235-244. [Crossref] [PubMed]
- Verlaan JJ, Diekerhof CH, Buskens E, et al. Surgical treatment of traumatic fractures of the thoracic and lumbar spine: a systematic review of the literature on techniques, complications, and outcome. Spine (Phila Pa 1976) 2004;29:803-14. [Crossref] [PubMed]
- Furlan JC, Noonan V, Cadotte DW, et al. Timing of decompressive surgery of spinal cord after traumatic spinal cord injury: an evidence-based examination of pre-clinical and clinical studies. J Neurotrauma 2011;28:1371-99. [Crossref] [PubMed]
- McCormack T, Karaikovic E, Gaines RW. The load sharing classification of spine fractures. Spine 1994;19:1741-4. [Crossref] [PubMed]
- Tator CH, Koyanagi I. Vascular mechanisms in the pathophysiology of human spinal cord injury. J Neurosurg 1997;86:483-92. [Crossref] [PubMed]
- Phang I, Werndle MC, Saadoun S, et al. Expansion duroplasty improves intraspinal pressure, spinal cord perfusion pressure, and vascular pressure reactivity index in patients with traumatic spinal cord injury: injured spinal cord pressure evaluation study. J Neurotrauma 2015;32:865-74. [Crossref] [PubMed]
- McKinley W, Meade MA, Kirshblum S, et al. Outcomes of early surgical management versus late or no surgical intervention after acute spinal cord injury. Arch Phys Med Rehabil 2004;85:1818-25. [Crossref] [PubMed]
- Dimar JR, Glassman SD, Raque GH, et al. The influence of spinal canal narrowing and timing of decompression on neurologic recovery after spinal cord contusion in a rat model. Spine 1999;24:1623-33. [Crossref] [PubMed]
- Cengiz SL, Kalkan E, Bayir A, et al. Timing of thoracolomber spine stabilization in trauma patients; impact on neurological outcome and clinical course. A real prospective (rct) randomized controlled study. Arch Orthop Trauma Surg 2008;128:959-66. [Crossref] [PubMed]
- Fehlings MG, Vaccaro A, Wilson JR, et al. Early versus Delayed Decompression for Traumatic Cervical Spinal Cord Injury: Results of the Surgical Timing in Acute Spinal Cord Injury Study (STASCIS). PLoS One 2012;7. [Crossref] [PubMed]
- Wilson JR, Singh A, Craven C, et al. Early versus late surgery for traumatic spinal cord injury: the results of a prospective Canadian cohort study. Spinal Cord 2012;50:840-843. [Crossref] [PubMed]
- Copes WS, Champion HR, Sacco WJ, et al. The Injury Severity Score revisited. J Trauma 1988;28:69-77. [Crossref] [PubMed]
- International Standards for Neurological Classification of Spinal Cord Injury. Chicago, IL: American Spinal Injury Association, 2000.
- Bracken MB, Shepard MJ, Holford TR, et al. Administration of methylprednisolone for 24 or 48 hours or tirilazad mesylate for 48 hours in the treatment of acute spinal cord injury. Results of the Third National Acute Spinal Cord Injury Randomized Controlled Trial. National Acute Spinal Cord Injury Study. JAMA 1997;277:1597-604. [Crossref] [PubMed]
- Mazaira J, Labanda F, Romero J, et al. Epidemiología de la lesión medular y otros aspectos. Rehabilitación 1998;32:365-72.
- Harris MB, Sethi RK. The initial assessment and management of the multiple-trauma patient with an associated spine injury. Spine (Phila Pa 1976) 2006;31:S9-15. [Crossref] [PubMed]
- Hurlbert RJ, Hadley MN, Walters BC, et al. Pharmacological therapy for acute spinal cord injury. Neurosurgery 2013;72:93-105. [Crossref] [PubMed]
- Batchelor PE, Wills TE, Skeers P, et al. Meta-analysis of pre-clinical studies of early decompression in acute spinal cord injury: a battle of time and pressure. PLoS One 2013;8. [Crossref] [PubMed]
- Pizones J, Izquierdo E, Sánchez-Mariscal F, et al. Sequential Damage Assessment of the Different Components of the Posterior Ligamentous Complex After Magnetic Resonance Imaging Interpretation: Prospective Study 74 Traumatic Fractures. Spine (Phila Pa 1976) 2012;37:E662-7. [Crossref] [PubMed]
- Perkins PG, Deane RH. Long-term follow-up of six patients with acute spinal injury following dural decompression. Injury 1988;19:397-401. [Crossref] [PubMed]
- Harrop JS, Naroji S, Maltenfort MG, et al. Neurologic improvement after thoracic, thoracolumbar, and lumbar spinal cord (conus medullaris) injuries. Spine (Phila Pa 1976) 2011;36:21-5. [Crossref] [PubMed]
- Carreon LY, Dimar JR. Early versus late stabilization of spine injuries: a systematic review. Spine (Phila Pa 1976) 2011;36:E727-733. [Crossref] [PubMed]
- Pointillart V, Petitjean ME, Wiart L, et al. Pharmacological therapy of spinal cord injury during the acute phase. Spinal Cord 2000;38:71-6. [Crossref] [PubMed]
- La Rosa G, Conti A, Cardali S, et al. Does early decompression improve neurological outcome of spinal cord injured patients? Appraisal of the literature using a meta-analytical approach. Spinal Cord 2004;42:503-512. [Crossref] [PubMed]
- Fehlings MG, Perrin RG. The timing of surgical intervention in the treatment of spinal cord injury: a systematic review of recent clinical evidence. Spine (Phila Pa 1976) 2006;31:S28-35. [Crossref] [PubMed]