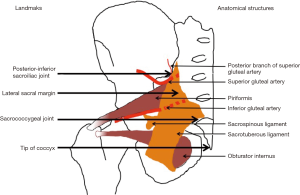
Identifying the superior and inferior gluteal arteries during a sacrectomy via a posterior approach
Introduction
Sacral tumors pose a challenging surgical problem due to their relative rareness and complex regional anatomy (1,2). Multiple surgical techniques have been described, but often need to be modified to account for the unique patient and tumor characteristics. A posterior approach is typically utilized, however, the anatomy, particularly the identification of anterior structures from a posterior approach, is often unfamiliar and challenging (2-5).
Of particular concern is the identification and control of gluteal vessels, which can pose acute life-threatening hemorrhage or delayed flap necrosis and subsequent deep infection if injured (1,6-10). In the perisacral region, these vessels are covered by the sacrospinous and sacrotuberous ligaments, which obscure their identification during a posterior approach (11).
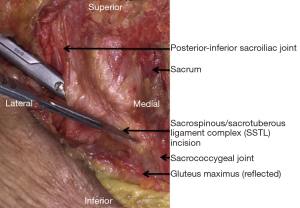
The sacrospinous ligament is a triangular shaped structure that originates on the anterior sacrum and coccyx and inserts on the ischial spine (12). Medially, its fibers are intertwined with the more posterior fan-shaped sacrotuberous ligament, which originates on the sacrum, coccyx, ilium and sacroiliac joint capsule and inserts on the ischial tuberosity (12,13). Because the fibers of these ligaments are conjoined medially, this region is described as the sacrospinous/sacrotuberous ligament complex (SSTL).
The SSTL overlies the gluteal vessels, making it difficult to determine their position prior to incising the SSTL. Vascular injury can therefore occur in the process of releasing and reflecting the SSTL, making an accurate understanding of the vascular position prior to its incision advantageous (1). Furthermore, with the advances in surgical techniques, particularly minimizing lateral dissection, an accurate understanding of the vascular position is necessary if less invasive techniques are to be utilized.
The location, course and distribution of the superior and inferior gluteal vessels are well documented, particularly in relation to the sciatic notch, anterior pelvic surgery and posterior approach to the hip (11,14,15). However, to our knowledge, there are no previous publications on the relative position of the SGA and IGA to defined anatomical landmarks to aid surgeons in their identification during a posterior sacrectomy. Therefore, this study aims to define useful palpable landmarks that can be used to approximate the location of these vessels during a posterior sacrectomy.
Methods
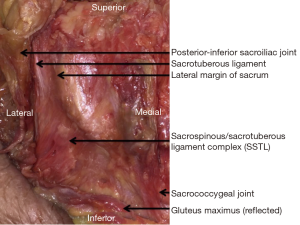
After ethical approval was obtained from the local anatomy ethics committee, six fresh human cadavers (3 male, 3 female, age range 80–101 years) were harvested. The left and right perisacral regions were analyzed independently, generating 12 samples. Cadavers were positioned prone in the anatomical position and the lateral margin of the sacrum, the posteroinferior aspect of the sacroiliac joint (PISIJ), the apex of the coccyx and the sacrococcygeal joints (SCJ) were palpated and marked. A posterior dissection of the sacrum and perisacral tissues was then performed through a direct midline incision with proximal-lateral extension as required.
After the skin incision, the lateral margin of the sacrum, PISIJ, apex of the coccyx and SCJ were palpated through the wound and marked. The gluteus maximus was then cleared of overlying subcutaneous tissue and elevated from medial to lateral to expose the SSTL, PISIJ, SCJ and apex of the coccyx. The lateral sacral margin was palpated.
The SSTL was then longitudinally incised 1cm from the lateral sacral margin and reflected laterally. The piriformis, SGA and IGA were identified. The position of the vessels, 1 cm lateral to the lateral sacral margin, in relation to the landmarks was measured with a caliper. Medial and lateral dissection of the vessels was then performed to analyze their position and course relative to the landmarks.
Finally, the width of the sacrum was measured at the level of the PISIJ and SCJ. A horizontal sacral osteotomy was performed, at the level of the PISIJ, and the level of this landmark in relation to the anterior sacral foramina determined.
Results
The SCJ, apex of coccyx and lateral sacral margin were easily palpable landmarks throughout the procedure. The PISIJ was more difficult to palpate prior to the incision, because the gluteus maximus covers this landmark. The position was therefore estimated by the confluence of the ilium and the lateral sacral margin. After the skin incision, palpation of the PISIJ through the gluteus maximus was easy and accurate.
In all samples the sacrotuberous component of the SSTL extended proximally above the PISIJ (average 32 mm, range 29–37 mm). However, at the level of the PISIJ, the SSTL over-hung the lateral sacrum by only 4.5 mm (range, 4–5 mm). More distally the SSTL extended laterally as a broad ligament complex towards their insertions on the ischial spine and ischial tuberosity. The distal origin of the SSTL extended to the SCJ (Figure 1).
The position of the PISIJ was most commonly at the level of the S3 anterior foramen but varied between individuals (range S2/3–S3/4). The width of the sacrum at this level varied (average 82 mm, range 72–88 mm). There were anatomical variations identified between each sacrum with the width at the level of the SCJ averaging 26 mm (range, 21–30 mm).
The SGA was found within 5 mm of the PISIJ in all but one cadaver (Figure 2). In all samples, the SGA was tethered to this region by a constant posterior branch. This posterior vessel coursed immediately inferior to the PISIJ before branching to supply the soft tissues around the inferior SIJ (Figure 3).
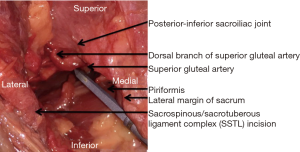
In the atypical cadaver, the PISIJ had the lowest position (S3/4) and the SGA was found 15 mm (left side) and 20 mm (right side) cephalad of the PISIJ.
The depth of the SGA relative to the SSTL, 1cm lateral to the lateral sacral margin was on average 13 mm (range, 5–17 mm). This became deeper more medially as the vessel coursed towards its origin on the internal iliac artery. At the level of the lateral sacrum the depth was on average 24 mm (range, 15–40 mm). More laterally a perforator of the SSTL was found in all cases, which tethered the SGA to the SSTL.
The IGA had a similar course. At 1 cm lateral to the lateral sacral margin, it was found on average 28 mm (range, 24–39 mm) caudal of the PISIJ. This corresponded to the midpoint between the PISIJ and SCJ, which was essentially the midpoint of the SSTL. Its depth relative to the SSTL, at this point, was on average 18 mm (range, 14–30 mm). This became deeper the more medial we dissected as the vessel coursed towards its origin on the internal iliac artery. At the level of the lateral sacrum the depth was on average 24 mm (range, 15–30 mm). More laterally a perforator of the SSTL was found in 10/12 samples, which tethered the IGA to the SSTL.
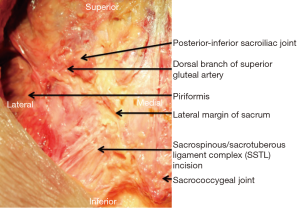
In all specimens we found a clear anatomical plane anterior of the SSTL, within 2 cm of the lateral sacral margin, which could be used to protect the vessels during SSTL resection (Figure 4).
Discussion
Perisacral surgery is challenging, particularly identifying and protecting anterior structures from a posterior approach (1,2,15). Wide dissection was historically utilized to aid identification of anatomical structures, but this causes significant soft tissue disruption (15). Thus, more recently, less invasive techniques have been favored if the pathology permits. These techniques limit lateral dissection or utilize percutaneous techniques to release the SSTL from the sacrum.
Irrespective of the type of surgery, in a posterior sacrectomy, the SSTL acts as a useful landmark to the depth of dissection and is used to prevent plunging into anterior structures, notably the gluteal vessels. However, the identification of the SGA and IGA is obstructed by the SSTL, which covers these vessels in the perisacral region. Its incision, which is necessary to release the sacrum, can therefore inadvertently injure these vessels (1). Surgical techniques, to identify these vessels prior to SSTL release are therefore necessary (16).
This study has identified key anatomical landmarks to help surgeons predict the location of these vessels during the perisacral release (Figure 5). The tip of the coccyx, SCJ and lateral sacrum are easily palpable landmarks throughout the procedure. The PISIJ is more difficult to palpate prior to the incision, but once the skin is incised, palpation of this landmark through the gluteus maximus is easy and accurate.
Once these landmarks are identified the position of the vessels can be accurately estimated. In practice, because the SSTL offers protection to the gluteal vessels we commence the partial elevation of gluteus maximus from the SSTL in the midpoint between the PISIJ and the SCJ. Then proximal dissection and palpation can easily identify the superior border of the SSTL as a thick rolled edge. The superior edge is then cleared and dissected cephalad towards the PISIJ. The consistent posterior branch of the SGA is located immediately superolateral to the SSTL at the inferior extent of the SIJ, thus staying along the superior border of the SSTL affords protection from damaging this vessel.
The SGA is typically found immediately inferior to the PISIJ and 5 mm or more anterior to the SSTL. It is tethered to this region by its posterior branch, which wraps around the inferior SIJ. This branch may bleed profusely if injured during the procedure and care should therefore be taken when dissecting in this region. However, once identified, the posterior branch can be used to identify the SGA, by dissecting anteriorly along its course.
Distally a similar approach is used to elevate the gluteus maximus off the SSTL and affords easy identification of the inferior border of the SSTL. At the level of the lateral sacrum, the IGA is typically positioned in the midpoint of the SSTL or midpoint between the PISIJ and SCJ. This corresponds to around 3 cm distal to the PISIJ, but this varies with the relative position of the PISIJ on the sacrum.
Because a clear anatomical plane exists anterior to the SSTL a blunt instrument can be inserted along its anterior surface to protect anterior structures, including the gluteal vessels, during its release (Figure 4). However, care should be taken with lateral reflection of this ligament complex, because perforators of both the SGA and IGA traverse the SSTL. Once the SSTL is released the piriformis and gluteal vessels can be easily identified. The SGA and IGA are more easily found laterally along the superior and inferior piriformis respectively, but more easily freed from the piriformis medially where the vessels are typically surrounded by fat and a clear plane can be created between the muscle and vessels.
It is important to recognize that both vessels move more anteriorly the more medially one dissects, and this suggests that the more medial the SSTL can be released the more separated the vessels are likely to be from the anterior border of this ligament complex. Advantageously, this is sometimes exaggerated with an anterior tumor mass, which displaces the vessels anteriorly.
This study is limited by the use of cadavers without sacral pathology. Clearly, the tumor can distort the local anatomy (1). Furthermore, preoperative radiotherapy can result in local scarring, including to the gluteal vessels (1). Therefore, intraoperatively, the parameters described in this study may be affected.
Another limitation is the low numbers, which cannot account for the variance in the SSTL, SGA and IGA as well as the level of the PISIJ relative to the anterior sacral foramina (12,17). On average, the PISIJ was found to be at the level of the S3 anterior sacral foramen. In practice, this can be easily determined on preoperative cross-sectional imaging, which can be used to aid the intra-operative determination of the proximal sacral osteotomy. Furthermore, understanding that the piriformis muscle originates from the middle three segments of the sacrum and that the gluteal vessels run along its superior and inferior border, allows the surgeon to predict the position of these vessels relative to the PISIJ, but only if they are not clearly identified on the preoperative images (11). Alternatively, preoperative angiography or intraoperative Doppler ultra-sound may be used to identify the vessels.
In conclusion, the posterior-inferior aspect of the sacroiliac joint, lateral sacral margin and SCJ are reliable anatomical landmarks that can be used to predict the location of the gluteal vessels during a posterior sacrectomy. An anatomical plane exists anterior to the sacrospinous/sacrotuberous ligament complex, which provides protection of these vessels during its transection. Furthermore, the distance between the vessels and this ligament complex increases the closer it is released to the lateral sacral margin.
Acknowledgements
The authors thank Glynny Kieser for her editorial input.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: Ethical approval was obtained from the local anatomy ethics committee. Approval was deemed part of the grouped approval for spinal cadaveric research (approval ID: NA).
References
- kiyama T. Anatomical difficulties of hemipelvectomy and sacrectomy from the view of vascular complex. Anat Physiol: Cur Res 2017;7:268-9.
- Karakousis C, Sugarbaker P. Sacrectomy. In: Malawer MM, Sugarbaker PH. editors. Musculoskeletal cancer surgery. Dordrecht: Kluwer Academic Publishers, 2001:415-24.
- Randall RL, Bruckner J, Lloyd C, et al. Sacral resection and reconstruction for tumors and tumor-like conditions. Orthopedics 2005;28:307-13. [PubMed]
- Tomita K, Tsuchiya H, Kawahara N, et al. En bloc sacrectomy. In: Watkins RG, Watkins RG. editors. Surgical approaches to the spine. New York: Springer, 2015;37:245-50.
- Zhang HY, Thongtrangan I, Balabhadra RS, et al. Surgical techniques for total sacrectomy and spinopelvic reconstruction. Neurosurg Focus 2003;15:E5. [Crossref] [PubMed]
- Altman DT, Jones CB, Routt ML Jr. Superior gluteal artery injury during iliosacral screw placement. J Orthop Trauma 1999;13:220-7. [Crossref] [PubMed]
- Belley G, Gallix BP, Derossis AM, et al. Profound hypotension in blunt trauma associated with superior gluteal artery rupture without pelvic fracture. J Trauma 1997;43:703-5. [Crossref] [PubMed]
- Lim EV, Lavadia WT, Roberts JM. Superior gluteal artery injury during iliac bone grafting for spinal fusion. A case report and literature review. Spine (Phila Pa 1976) 1996;21:2376-8. [Crossref] [PubMed]
- Senchenkov A, Moran SL, Petty PM, et al. Predictors of complications and outcomes of external hemipelvectomy wounds: account of 160 consecutive cases. Ann Surg Oncol 2008;15:355-63. [Crossref] [PubMed]
- Sim FH, Choong PF, Weber KL. Orthopaedic oncology and complex reconstruction. In: Sim FH. editor. Master techniques in orthopaedic surgery. Philadelphia: Lippincott Williams & Wilkins, 2010;6:3-82.
- McMinn RM. Lasts anatomy: Regional and applied. 9th ed. Churchill Livingstone, 2003:164.
- Hammer N, Steinke H, Slowik V, et al. The sacrotuberous and the sacrospinous ligament--a virtual reconstruction. Ann Anat 2009;191:417-25. [Crossref] [PubMed]
- Loukas M, Louis RG Jr, Hallner B, et al. Anatomical and surgical considerations of the sacrotuberous ligament and its relevance in pudendal nerve entrapment syndrome. Surg Radiol Anat 2006;28:163-9. [Crossref] [PubMed]
- Collinge CA, Ziran NM, Coons DA. Relationship Between the Superior Gluteal Vessels and Nerve at the Greater Sciatic Notch. Orthopedics 2015;38:e929-33. [Crossref] [PubMed]
- Zoccali C, Skoch J, Patel A, et al. The surgical neurovascular anatomy relating to partial and complete sacral and sacroiliac resections: a cadaveric, anatomic study. Eur Spine J 2015;24:1109-13. [Crossref] [PubMed]
- Akiyama T, Kanda S, Maeda A, et al. Inferior gluteal artery detection at the greater sciatic notch with a computer-assisted navigation system during pelvic and sacral tumor resection. Comput Assist Surg (Abingdon) 2016;21:18-24. [Crossref] [PubMed]
- Lai J, du Plessis M, Wooten C, et al. The blood supply to the sacrotuberous ligament. Surg Radiol Anat 2017;39:953-9. [Crossref] [PubMed]