Three-dimensional reconstructions in spine and screw trajectory simulation on 3D digital images: a step by step approach by using Mimics software
Introduction
Understanding bony anatomy is of paramount importance for spine surgeons. One way to study bony anatomy for surgical technique training and testing of innovative instrumentation is via cadaveric specimens, these are often limited in availability. In the majority of countries, the availability of cadaveric specimens for testing remains limited given the low donation rates (1).
One alternative avenue for study of surgical anatomy and testing of novel fixation designs is via three-dimensional (3D) reconstructed images and models (2-4). 3D reconstructed images can be derived from original computed tomography (CT) or magnetic resonance imaging (MRI), and studies have shown these can be sufficiently used for morphometric study, and have advantages of being accurate, “clean” and reusable (5,6), or for further 3D-printed implants (7,8).
However, the despite the increasing use of 3D reconstructive technologies in the literature, there is limited step-by-step protocols available to guide the surgeon to perform such 3D reconstructions in the office setting. Furthermore, there is no routine consensus on the ideal method of 3D reconstruction from CT and MRI imaging, which limits the reliability and dissemination of results amongst surgeons and the surgical community. To address these limitations, this is the first article of a three-article series outlining a step-by-step protocol which will allow readers to easily and reliably produce 3D reconstructions in their further research, assist with the study of bony anatomy and the design of novel screw fixation techniques or pre-operative surgical planning.
Step-by-step approach of 3D reconstruction
- First of all, it is necessary to obtain continuous CT images in format of DICOM, and saved in your local drive. In our institution, this can be downloaded from the PACS system in our hospital. Before you download the CT data, a written informed consent should be obtained from participant and your research project should be approved by your institutional ethics review board (IRB). The Mimics software (Materialise NV, Leuven, Belgium) is onto your computer.
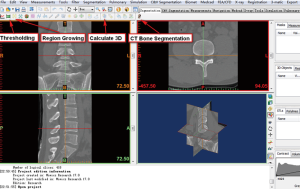
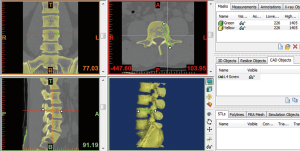
- Open the Mimics software. Click the upper left corner button: File—New project wizard—select your own DICOM data (your data saved route)—Next—wait for all of the data imported—Convert—wait for software conversion—the software will convert the DICOM data to Figure 1.
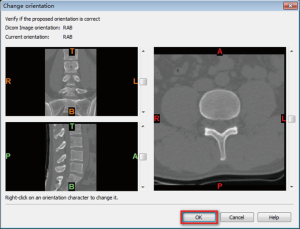
- After confirmation of the orientation character [T (top), B (bottom), A (anterior), P (posterior), L (left), R (right)], click “OK”, go to the Figure 2 interface.
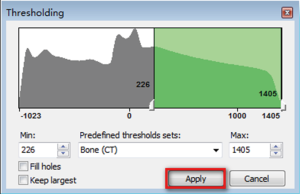
- Click the button “Thresholding”, which display the following dialog box (Figure 3). We want reconstruct the bone structure, therefore, the predefined thresholds sets was selected as “Bone (CT)”, the default thresholding value of Bone (CT) is range from “226—a certain value (depends on the bone mineral density of participant)”. The researcher can also choose other values according to their research condition. If the “Min” value is too lower, it will be hard to separate different bones. If the “Min” value is too higher, there will be many bony defect areas in the 3D reconstructed bone. After choosing an appropriate thresholding value, click “Apply”, the “Region Growing Toolbar” (Figure 4) will be displayed.
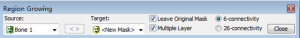
- We can see the target “New Mask” at the Figure 4, and then click the reconstructed interested area on Figure 5. Here, we want to reconstruct the bone structure of lumbar spine, so we click on the vertebral bone area, the interested region will be turned into the target color as Figure 4 (yellow color here). Any structure that is connected with the pre-selected thresholding value—“226–1405” will be regarded as one entire object, and turned to be target color.
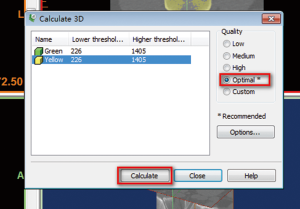
- Click the “Calculate 3D” button as Figure 2 shows. The dialog box of “Calculate 3D” (Figure 6) will be displayed. We select the mask of “Yellow”, and the quality of “Optimal”. Researchers can choose the other options depending on their needs or preferences. The researchers also can skip Region Growing steps, directly use the original “Green” mask for 3D reconstruction if they only reconstruct the entire bone, without the separate the different bones. Here, we attempt to use “Region Growing” function to show the readers that may be some isolated bone can be separated from other bone. Then, click on the “Calculate” to calculate the 3D image. The reconstructed 3D image will be calculated and showed on the lower right frame of Figure 5, and saved it on your local computer for further research.
Step by step approach of creating screw trajectory simulation on 3D digital images
- We can use the above reconstructed 3D image or use previously saved reconstructed 3D images. If using a reconstructed 3D lumbar spine, firstly, we open the Mimics software, then, click the upper left “File”—select “Open project”—find your file save route—click “Open” to open your 3D lumbar spine.
- Now, we want to create one pedicle screw trajectory on the third lumbar segment of 3D lumbar image. Then, we choose the “MedCAD” module (Figure 7)—select the “Create Cylinder”—at this time, the cursor will be changed to be a pen—move the cursor to 3D object (third lumbar vertebra) and determine a start point at 3D object as the end of screw trajectory, and click the left button of the mouse to generate the start point—then move the cursor to the point of another end of screw trajectory and click the left button of the mouse again to generate the second point—at this time the length of your screw trajectory was determined, but not the radius—then, move your cursor side to determine your radius and click the left button of the mouse again—a cylinder is now generated to simulate the screw trajectory. However, the screw trajectory created is currently crude and not your desired one, and still requires adjustment and modification in the next step.
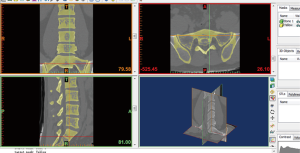
- The position of screw trajectory could be adjusted at the sagittal plane, coronal plane, axial plane or 3D object frame. When you click the screw trajectory at each above four frames, there will be displayed two white end points at the screw trajectory, and you can drag one of them to ideal place to adjust the screw trajectory. In the 3D object frame, the screw trajectory may be hidden inside of the 3D object, to visualize the screw trajectory; we can click on the button of “Toggle Transparency” (Figure 7) to change the transparency of 3D object.
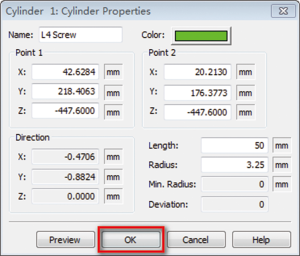
- The original radius of the screw trajectory may not meet our research requirements. Therefore to modify the radius of screw trajectory, we can click “CAD Objects” (Figure 7)—select the “cylinder” which represents the screw trajectory—click “Properties”—the dialog box of “Cylinder properties” will be displayed (Figure 8), you can change the radius, color and length of your cylinder at this dialog box (here, we change the radius =3.25 mm)—then click “OK” to close this dialog box.
- After setting up the satisfactory screw trajectory, you can observe the screw trajectory, measure the length and angles of screw trajectory for pre-operative planning or investigate some novel screw fixation techniques.
Discussion
In this protocol, we use Mimics software and CT data to reconstruct the 3D digital reconstructions of lumbar spine. The key interfaces were captured and listed, and all of the procedures were displayed step-by-step. After the 3D digital image of lumbar spine reconstruction, we choose the third lumbar vertebra and use “Cylinder” to simulate the pedicle screw trajectory.
The above step-by-step approach is only one of many possible 3D reconstruction methods. Mimics software is one of the many different 3D reconstruction softwares, others included Amira, 3D doctor, Simpleware, 3D Med, 3D Slicer (9,10). Mimics software has worldwide popularity amongst medical researchers for its convenience and quick 3D reconstructions from axial CT images.
Mimics works as bridge to connect the CT or MR images to further various research. The 3D reconstructed images can easily use for further finite element analysis (11,12). The accurate 3D reconstructed image also can be used for anatomic measurement. Qian et al. (13) use Mimics software to measure the lumbar pedicle-lengthening distance and estimate the lumbar stenosis before surgery, and Puchwein et al. use it to measure the odontoid peg and assess whether the odontoid peg can offer enough room for two 3.5-mm cannulated cortical screws(14). Izumi et al. (15) used it to measure the ossification of the posterior longitudinal ligament (OPLL) of the cervical spine, find it allowed detailed ossification of the OPLL classification and very useful for quantitative evaluation of OPLL with only minimal measurement error. Wu et al. (2) use the screw trajectory simulation function of Mimics software to study the feasibility of novel transdiscal screw fixation in lumbar spine, and design the trapezoid shape interbody cages for novel percutaneous posterior transdiscal oblique screw fixation with lateral interbody fusion technique (16).
In the 3D printing realm, the accurate 3D reconstructed digital images reconstructed by Mimics software can further imported to a 3D printer to print accurate bony models (17,18), which may replace the specimen of human body for medical education (19,20), and surgeon training (21) and without the ethical problem of human body donation (22). Lu et al. (4,23) used this method to design some novel rapid prototyping drill templates for cervical pedicle screw placement and C2 laminar screws, they suggest that these patient-specific templates are easy to use, fit the patient anatomy and can generate highly accurate screw placement.
Although above step-by-step approach to reconstruct the 3D digital image is simple, easily and useful method, it is by no means necessarily superior to other methods of achieving 3D reconstructions. The readers can use this method or others according to their personal preference, however, we advocate this protocol for its simplicity, reliability and convenience in obtaining the 3D digitally reconstructed images.
Acknowledgements
Funding: This work was supported by the National Natural Science Foundation of China (81501933), Zhejiang Provincial Medical Technology Foundation of China (2018KY129), Wenzhou Municipal Science and Technology Bureau (Y20170389), and Wenzhou Leading Talent Innovative Project (RX2016004).
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Boulware LE, Ratner LE, Cooper LA, et al. Whole body donation for medical science: a population-based study. Clin Anat 2004;17:570-7. [Crossref] [PubMed]
- Wu AM, Tian NF, Wu LJ, et al. A radiological and cadaveric study of oblique lumbar interbody fixation in patients with normal spinal anatomy. Bone Joint J 2013;95-B:977-82. [Crossref] [PubMed]
- Choy WJ, Mobbs RJ, Wilcox B, et al. Reconstruction of Thoracic Spine Using a Personalized 3D-Printed Vertebral Body in Adolescent with T9 Primary Bone Tumor. World Neurosurg 2017;105:1032.e13-17. [Crossref] [PubMed]
- Lu S, Xu YQ, Zhang YZ, et al. A novel computer-assisted drill guide template for placement of C2 laminar screws. Eur Spine J 2009;18:1379-85. [Crossref] [PubMed]
- Jamali AA, Deuel C, Perreira A, et al. Linear and angular measurements of computer-generated models: are they accurate, valid, and reliable? Comput Aided Surg 2007;12:278-85. [Crossref] [PubMed]
- Gelaude F, Vander Sloten J, Lauwers B. Accuracy assessment of CT-based outer surface femur meshes. Comput Aided Surg 2008;13:188-99. [Crossref] [PubMed]
- Mobbs RJ, Coughlan M, Thompson R, et al. The utility of 3D printing for surgical planning and patient-specific implant design for complex spinal pathologies: case report. J Neurosurg Spine 2017;26:513-8. [Crossref] [PubMed]
- Phan K, Sgro A, Maharaj MM, et al. Application of a 3D custom printed patient specific spinal implant for C1/2 arthrodesis. J Spine Surg 2016;2:314-8. [Crossref] [PubMed]
- Ni Y, Dai P, Dai C, et al. Cochlear implant-related three-dimensional characteristics determined by micro-computed tomography reconstruction. Clin Anat 2017;30:39-43. [Crossref] [PubMed]
- Hieu LC, Sloten J, Hung LT, et al. Medical reverse engineering applications and methods. 2ND International Conference on Innovations, Recent Trends and Challenges in Mechatronics, Mechanical Engineering and New High-Tech Products Development, MECAHITECH. Bucharest, 23-24 September 2010.
- Song M, Zhang Z, Lu M, et al. Four lateral mass screw fixation techniques in lower cervical spine following laminectomy: a finite element analysis study of stress distribution. Biomed Eng Online 2014;13:115. [Crossref] [PubMed]
- Lu S, Xu YQ, Zhang MC, et al. Biomechanical effect of vertebroplasty on the adjacent interver tebral levels using a three-dimensional finite element analysis. Chin J Traumatol 2007;10:120-4. [PubMed]
- Qian L, Li P, Wu W, et al. Restoration of the spinal canal volume in stenosis dependent on pedicle-lengthening distance in pedicle-lengthening osteotomy: A three dimensional simulation. Bone Joint J 2016;98-B:238-43. [Crossref] [PubMed]
- Puchwein P, Jester B, Freytag B, et al. The three-dimensional morphometry of the odontoid peg and its impact on ventral screw osteosynthesis. Bone Joint J 2013;95-B:536-42. [Crossref] [PubMed]
- Izumi T, Hirano T, Watanabe K, et al. Three-dimensional evaluation of volume change in ossification of the posterior longitudinal ligament of the cervical spine using computed tomography. Eur Spine J 2013;22:2569-74. [Crossref] [PubMed]
- Wu AM, Ni WF, Shao ZX, et al. Percutaneous posterior transdiscal oblique screw fixation with lateral interbody fusion: a radiological and cadaveric study. Eur Spine J 2015;24:852-8. [Crossref] [PubMed]
- Wu AM, Shao ZX, Wang JS, et al. The accuracy of a method for printing three-dimensional spinal models. PLoS One 2015;10:e0124291. [Crossref] [PubMed]
- Shui W, Zhou M, Chen S, et al. The production of digital and printed resources from multiple modalities using visualization and three-dimensional printing techniques. Int J Comput Assist Radiol Surg 2017;12:13-23. [Crossref] [PubMed]
- Lim KH, Loo ZY, Goldie SJ, et al. Use of 3D printed models in medical education: A randomized control trial comparing 3D prints versus cadaveric materials for learning external cardiac anatomy. Anat Sci Educ 2016;9:213-21. [Crossref] [PubMed]
- Jones DB, Sung R, Weinberg C, et al. Three-Dimensional Modeling May Improve Surgical Education and Clinical Practice. Surg Innov 2016;23:189-95. [Crossref] [PubMed]
- Waran V, Narayanan V, Karuppiah R, et al. Utility of multimaterial 3D printers in creating models with pathological entities to enhance the training experience of neurosurgeons. J Neurosurg 2014;120:489-92. [Crossref] [PubMed]
- Halou H, Chalkias A, Mystrioti D, et al. Evaluation of the willingness for cadaveric donation in Greece: a population-based study. Anat Sci Educ 2013;6:48-55. [Crossref] [PubMed]
- Lu S, Xu YQ, Chen GP, et al. Efficacy and accuracy of a novel rapid prototyping drill template for cervical pedicle screw placement. Comput Aided Surg 2011;16:240-8. [Crossref] [PubMed]